Question: Succinic anhydride yields the cyclic imide succinimide when heated with ammonium chloride at 200 ?C. Propose a mechanism for this reaction. Why do you suppose
Succinic anhydride yields the cyclic imide succinimide when heated with ammonium chloride at 200 ?C. Propose a mechanism for this reaction. Why do you suppose such a high reaction temperature is required?
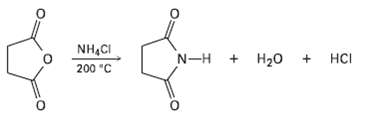
NH,CI HCI N-H H20 200 "C
Step by Step Solution
★★★★★
3.38 Rating (170 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

HC HC 30 02 HC HC 1 HC HNH3 H3O NH 9 HC O C HC OH OH ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
22-C-O-CA (150).docx
120 KBs Word File


