Butacetin is an analgesic (pain, killing) agent that is synthesized commercially from p-fluoronitrobenzene. Propose asynthesis. NHCOCH3 Butacetin
Question:
Butacetin is an analgesic (pain, killing) agent that is synthesized commercially from p-fluoronitrobenzene. Propose asynthesis.
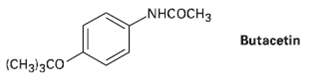
Transcribed Image Text:
NHCOCH3 Butacetin (CH3)3CO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
This synthesis requires a nucleophilic aromatic substitution r...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose structures for compounds O and P that are consistent with the following information. H2(2 equiv.C&12 0 DEPT CH2 CH (ppm) 13C NMR for Compound O 26.0 124.5
-
Commercially produced ice cream is made from a mixture of ingredients: A minimum of 10% milk fat 9 12% milk solids: this component, also known as the serum solids, contains the proteins (caseins...
-
An agent embezzles funds from his principal and uses the funds to buy a car. What duties has the agent violated? Who is entitled to ownership of the car?
-
Concentric shells r = 20 cm and r = 30 cm are held at V = 0 and V = 50, respectively. If the space between them is filled with dielectric material ( = 3.10, a = 1012 S/m), find: (a) V, E, and D, (b)...
-
How can you enhance your face-to-face conversations in the workplace and improve your image as a professional?
-
Flow occurs over a sharp-crested, uncontracted horizontal weir 3.5 ft high under a head of 1.0 ft. If this weir replaced another weir, which was one-half its height, what change in depth occurred in...
-
In Exercise 41, an error was made in grading your practical. Instead of getting 90, you scored 100. What is your new weighted mean? Data from Exercises 41 The scores and their percents of the final...
-
1. How long is a written stop-payment order effective? What else could RPM have done to prevent this check from being cashed? 2. What would happen if it turned out that RPM did not have a legitimate...
-
Glamly uses a normal costing system and applies manufacturing overhead to jobs at a rate of 110% of direct labor costs. The company had no beginning or ending raw materials or work-in-process...
-
Develop a unit test plan for a Web site that enables you to perform some function (e.g., make travel reservations, order books).
-
Succinic anhydride yields the cyclic imide succinimide when heated with ammonium chloride at 200 ?C. Propose a mechanism for this reaction. Why do you suppose such a high reaction temperature is...
-
Phenyl 4-aminosalicylate is a drug used in the treatment of tuberculosis. Propose a synthesis of this compound starting from 4-nitrosalicylicacid. CO2H H2N O2N Phenyl 4-aminosalicylate...
-
The sole issue to be decided in this appeal is the priority of competing liens in and to a manufactured [mobile] home located on, but not attached to, a parcel of real estate which is the subject of...
-
The following reaction was set-up as follows: R-OH HBr R-Br Where R-OH could be one of the following possible compounds 1-4
-
Solving the following questions below for the attach cased study: Questions: Briefly discuss the key drivers of the growth in green bonds. (4 marks) Recently, the green bond index has been...
-
4. You are given: (i) The number of claims follows a negative binomial distribution with parameters r and B = 3. (ii) Claim severity (Y) has the following distribution: Pr(Y = 1) = 0.5, Pr(Y = 10) =...
-
A solution of methyl acetate (C3HO) and water is boiling at 99.5 C. A sample of the vapor above the solution is cooled until it condenses. This condensed sample is analyzed, and turns out to be 10.%...
-
1. Consider the distributions: (i) Gamma: f(y:0) ==> (0) (o known). (ii) Negative binomial: f(y; 0) = (+1) 0 (1-0) (r known). For (i) and (ii): (a) Determine the maximum likelihood estimate, 0, of 0...
-
The following data relate to the characteristics of high-definition televisions A through E. Identify the individuals, variables, and data corresponding to the variables. Determine whether each...
-
In the current year, the City of Omaha donates land worth $500,000 to Ace Corporation to induce it to locate in Omaha and create an estimated 2,000 jobs for its citizens. a. How much income, if any,...
-
For each molecular geometry, list the number of total electron groups, the number of bonding groups, and the number of lone pairs on the central atom. (a) (b) (c)
-
Use the reaction mechanism, including the resonance structures of the carbocation intermediates, to predict the products of the following reactions. 1 ,3-butadience + HCl
-
Suggest a mechanism for each of the following reactions that accounts for both products. H,so, CH,CH CHCH,OH HBr CH,CH CHCH BrCH,CHCH CH2 (84%) Br (16%)
-
What would be the structure of polybutadiene if every other unit of the polymer resulted from 1,2-addition?
-
Explain the concept of hybrid thread models in operating systems. How do they attempt to combine the benefits of user-space and kernel-space threading ?
-
Describe the characteristics of the six stages of development for intimacy in a romantic relationship using a real or hypothetical example to illustrate the general developmental path.
-
Consider the following payoff matrix: Player 1 Strategy B Strategy A Strategy A (4,3) (6,2) Player 2 Strategy B (3,5) (2,4) Is the payoff matrix symmetric (Yes or No)?

Study smarter with the SolutionInn App


