Suggest syntheses of these amino acids from the indicated starting materials: NH, Br a) PHCH,CHCO,H from PHCH,CHCO,H
Question:
Suggest syntheses of these amino acids from the indicated starting materials:
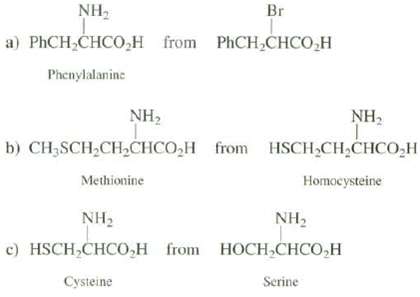
Transcribed Image Text:
NH, Br a) PHCH,CHCO,H from PHCH,CHCO,H Phenylalanine NH2 NH, b) CH3SCH2CHCHCO,H from HSCH CH,CHCO,H Methionine Homocysteine NH2 с) HSCH,CHCO,Н гom NH2 HОСH,CНCO,H Cysteine Serine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
Br a PhCHCHCOH excess NH3 NH ...View the full answer

Answered By

Marcus Solomon
I am committed to ensuring that my services always meet the clients' expectations.
4.60+
82+ Reviews
117+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show syntheses of these compounds from the indicated starting materials. More than one step may b necessary. Your syntheses may produce both enantiomers of any target that ischiral. , CH,CH,CH3 a)...
-
Propose syntheses of the following molecules from the indicated starting materials. Make use of any other reagents or solvents that you need. In some cases, there may be no alternative but to employ...
-
Show how Tagamet could be synthesized from the indicated starting materials. CH2OH CH CH SCH2CH2NH NHCH N CH CHS SCH CH3 +CH:NH2
-
Suggest two strategies for the company to minimise the impact of the strike on business operation?
-
In your own words, explain the difference between cost of goods manufactured and cost of goods sold.
-
Dale Thomas' rich uncle gave him $100,000 cash as a gift for his 40th birthday. Unlike his spoiled cousins who spend money carelessly, Mr. Thomas wants to invest the money for his future retirement....
-
What are the different types of organizational forms that corporations use for international operations?
-
Jason commenced with 135,000 cash. He acquired an established shop on 1 January 20X1. He agreed to pay 130,000 for the fixed and current assets and the goodwill. The replacement cost of the shop...
-
A manufacturing company reports the following Information for the month of May. Note: Assume all raw materials were used as direct materials. Activities for May Activities for May Advertising expense...
-
A device has been proposed that will serve as a blood oxygenator for a heartlung bypass machine, as shown in the figure below. In this process, blood containing no dissolved oxygen (O 2 , species A)...
-
Another Diphenhydramine synthesis is shown in the following equation: (a) Show a mechanism for the first step in this synthesis. (b) Explain which mechanism is occurring in the secondstep. OCH CH...
-
(a) Explain which of the following two products is formed when the reactant alkyl chloride reacts with sodium acetate in DMSO. (b) Explain which of the following two product alcohols is formed when...
-
In Problem find the logarithmic derivative. A(t) = 900e 0.24t
-
Explain what factors would typically be considered in contingency planning in outsourcing arrangements.
-
First-line managers are at the top of the managerial pyramid. a) True b) False
-
An affiliative leadership style is most useful when a radical change in corporate vision is needed. a) True b) False
-
What is the amount of net new financing needed for Jims?
-
Discuss the role played by product packaging in logistics systems.
-
Repeat Problem 14 for the system shown in Figure 5.50. Data From Problem 14: a. Draw the necessary free-body diagrams and derive the differential equations of motion. b. Write the differential...
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
Solve each equation. 8 2 =x + 3 5 + 23 11 -x+ 5 45 6x + 7 3
-
Pentalene is a most elusive molecule and has never been isolated. The pentalene dianion, however, is well known and quite stable. Explain. 12- Pentalene Pentalene dianion
-
Indole is an aromatic heterocycle that has a benzene ring fused to a pyrrole ring. Draw an orbital picture of indole. (a) How many ? electrons does indole have? (b) What is the electronic...
-
Ribavirin, an antiviral agent used against hepatitis C and viral pneumonia, contains a 1, 2, 4-triazole ring. Why is the ringaromatic? 1,2,4-Triazole ring N- NH2 N-N Ribavirin OH
-
Why was Thomas Paine's "common sense" article significant to the american revolution?
-
Aramex, Inc is currently offering a bond with a par value of $1,000, which is priced at $1,488 in the market. The bond offers a fixed annual coupon rate of 14.05 percent, which is paid semiannually....
-
The cash flow for the firm's project is -$40 million in year 0 and $19 million in years 1-4. After year 4, the FCF is expected to grow at a constant rate of 0.020. The firm's discount rate is 0.068....

Study smarter with the SolutionInn App


