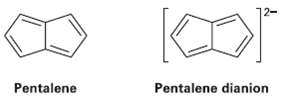
Pentalene is a most elusive molecule and has never been isolated. The pentalene dianion, however, is well
Question:
Pentalene is a most elusive molecule and has never been isolated. The pentalene dianion, however, is well known and quite stable. Explain.
Transcribed Image Text:
12- Pentalene Pentalene dianion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
Pentalene has eight elect...View the full answer

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The HCI molecule is quite well described by the Morse potential with De = 5.33 eV, V = 2989.7 cm-1, and XV = 52.05 cm-I. Assuming that the potential is unchanged on deuteration, predict the...
-
A South Korean research group has isolated and synthesized "daumone," the pheromone that induces hibernation in Caenorhabditis elegans worms when food becomes scarce, thus extending their life span...
-
The resonance form shown is not the most stable one for the compound indicated. Write the most stable resonance form.
-
Harvold Company's quality cost report is to be based on the following data: Test and inspection of incoming materials. $71,000 Supplies used in testing and inspection . Re-entering data because of...
-
Think of a positive customer experience. What are the behavioral success factors that made the encounter so successful?
-
In Section 2.6 it was noted that the net bonding energy EN between two isolated positive and negative ions is a function of interionic distance r as follows: where A, B, and n are constants for the...
-
With reference to Exercise 11.65, construct a \(99 \%\) confidence interval for \(\alpha\).
-
La Jolla Beverage Products is considering producing a wine cooler that would be a blend of a white wine, a ros wine, and fruit juice. To meet taste specifications, the wine cooler must consist of at...
-
Describe a time when you had to stop a client's violent or destructive behaviour. What did you do? What would you have done differently?
-
At December 31, 2010, Westport Manufacturing Co. owned the following investments in the capital stock of publicly owned companies (all classified as available-for-sale securities): In 2011, Westport...
-
Calicene, like azulene (Problem 15.17), has an unusually large dipole moment for a hydrocarbon. Explain, using resonancestructures. Calicene
-
Indole is an aromatic heterocycle that has a benzene ring fused to a pyrrole ring. Draw an orbital picture of indole. (a) How many ? electrons does indole have? (b) What is the electronic...
-
Scholars have made strong arguments for required representation on boards by stakeholders beyond stockholders such as employees, community members, and others, depending on the industry. What might...
-
a. Determine the ditribution of shear stress in the following circular shafts that are subject to a maximum torque of 7.5 Nm by calculating the polar second moment of area. a) b) a) O b) O b. Using...
-
Evaluate Nike's specific initiatives in the metaverse. What are the pros and cons of each? How can you help achieve company objectives? 2.How should these initiatives be organized, and which should...
-
Choose a company brand (based within the United States) that has been in business for a minimum of 20 years. Research the company based on the following components, which should be used as headings...
-
Below is a summary of some of ABC Corporation's transactions carried out during 2022, its first quarter of operations that ended on March 31. 1. ABC shareholders invested $300,000 in cash in exchange...
-
As the firm is cutting down on its capital expenditures, should it invest heavily in existing technology or in the stores themselves? What are the benefits and risks of either focus?
-
Upon graduation, Steven purchases a new home theater system for his apartment. To finance the system, he borrows \($5\),000 from a new credit card at 21 percent per year compounded monthly. He fully...
-
What are the key elements of a system investigation report?
-
Consider the chemical equation and equilibrium constant for the synthesis of ammonia at 25 C: Calculate the equilibrium constant for the following reaction at 25 C: N(g) + 3 H(g) = 2 NH3(8) K = 5.6 X...
-
Glutathione (GSH) is a tripeptide that serves as a mild reducing agent to detoxify peroxides and maintain the cysteine residues of hemoglobin and other red blood cell proteins in the reduced state....
-
Complete hydrolysis of an unknown basic decapeptide gives Gly, Ala, Leu, Ile, Phe, Tyr, Glu, Arg, Lys, and Ser. Terminal residue analysis shows that the N terminus is Ala and the C terminus is Ile....
-
There are many methods for activating a carboxylic acid in preparation for coupling with an amine. The following method converts the acid to an N-hydroxysuccinimide (NHS) ester. (a) Explain why an...
-
Blair, CPA, uses the cash receipts and disbursements method of reporting. In Year 7, a client gave Blair 100 shares of a listed corporation's stock in full satisfaction of a $5,000 accounting fee the...
-
What is driving the increase in the price of healthcare services today? Explain why it is important to know the scope of business being reviewed when using financial statements. Explain the content...
-
What is a discount? How do you activate discounts? Activate discounts: Assign a discount account: 3. Discounts Given - What is the account type and detail type for this account?

Study smarter with the SolutionInn App


