Question: Superior Micro Products uses the FIFO method in its process costing system. Data for the Assembly Department for May appear below: Required: Compute the cost
Superior Micro Products uses the FIFO method in its process costing system. Data for the Assembly Department for May appear below:
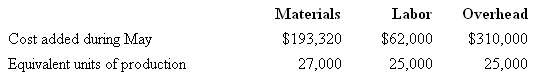
Required:
Compute the cost per equivalent unit for materials, labor, overhead, and intotal.
Materials Labor Overhead Cost added during May Equivalent units of production $193,320 $62,000 25,000 $310,000 25,000 27,000
Step by Step Solution
3.48 Rating (165 Votes )
There are 3 Steps involved in it
FIFO method Materials Labor Overhead Cost added during May a 193320 ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
25-B-M-A-C-S-D (82).docx
120 KBs Word File


