Suppose that the silver-silver chloride electrode in Figure 14-2 is replaced by a saturated calomel electrode. Calculate
Question:
Suppose that the silver-silver chloride electrode in Figure 14-2 is replaced by a saturated calomel electrode. Calculate the cell voltage
if [Fe2+] / [Fe3+] = 2.5 × 10-3.
Figure 14-2
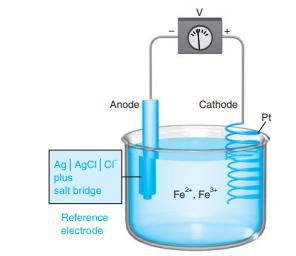
Transcribed Image Text:
Anode Cathode Pt Ag AgCi|cr plus salt bridge Fe", Fe* Reference electrode
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
E E E E 077...View the full answer

Answered By

Payal Mittal
I specialize in finance and accounts.You can ask any question related to til undergradution.Organizational behaviour and HRM are my favourites for you can always relate to them and is an art with practical knowledge base.
4.90+
226+ Reviews
778+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
For a silver-silver chloride electrode, the following potentials are observed: E = 0.222 V E(saturated KCl) = 0.197 V From these potentials, find the activity of Cl - in saturated KCl. Calculate E...
-
A voltaic cell is constructed with two silver-silver chloride electrodes, each of which is based on the following halfreaction: AgCl(s) + e- Ag(s) + Cl- (aq) The two half-cells have [CI-] = 0.0150 M...
-
Suppose that the Ag | AgCl outer electrode in Figure 14-11 is filled with 0.1 M NaCl instead of saturated KCl. Suppose that the electrode is calibrated in a dilute buffer containing 0.1 M KCl at pH...
-
The reconciling item in a bank reconciliation that will result in an adjusting entry by the depositor is: (a) outstanding checks. (b) deposit in transit. (c) a bank error. (d) bank service charges.
-
For the year ended December 31, 2014, Denkinger Electrical Repair Company reports the following summary payroll data. Gross earnings: Administrative salaries ............$200,000 Electricians wages...
-
ITown is a large computer discount store that sells computers and ancillary equipment and software in the town where State University is located. It has collected historical data on computer sales...
-
Among your colleagues in class, identify a term or phrase italicized in this chapter that you think is the most significant from your reading. Absent team consensus, then just provide your...
-
Nathan Industries had a remaining debit balance of $20,000 in its under-and overapplied factory overhead account at year-end. It also had year-end balances in the following accounts: Work in...
-
Lopez Sales Company had the following balances in its accounts on January 1 , Year 2 : Cash $ 6 2 , 0 0 0 Merchandise Inventory 4 2 , 0 0 0 Land 1 0 2 , 0 0 0 Common Stock 8 2 , 0 0 0 Retained...
-
IBS is a global provider of point-of-sale systems and related services that enable businesses to accept electronic payments. As a new hire in the companys international headquarters accounting...
-
What does the selectivity coefficient tell us? Is it better to have a large or a small selectivity coefficient?
-
Why is it preferable to use a metal ion buffer to achieve pM = 8 rather than just dissolving enough M to give a 10-8 M solution?
-
A doctor is asked to give an executive a thorough physical checkup to test the null hypothesis that he will be able to take on additional responsibilities. Explain under what conditions the doctor...
-
Lisinipril is a drug designed to lower blood pressure. In a clinical trial of Lisinipril, blood pressure levels of subjects are measured before and after they have been treated with the drug....
-
Identifying the Population In a Gallup poll of 1010 adults in the United States, 55% of the respondents said that they used local TV stations daily as a source of news. Is the 1010 value a statistic...
-
According to the State of New York Unified Court System, names of potential jurors are selected from a variety of different sources. When a trial requires a jury, names from the list are randomly...
-
Years in which U.S. presidents were inaugurated. Determine which of the four levels of measurement (nominal, ordinal, interval, ratio) is most appropriate.
-
When collecting data from different sample locations in a lake, a researcher uses the line transect method by stretching a rope across the lake and collecting samples at every interval of 5 meters....
-
The pressure on a sample of hydrogen is doubled, while its temperature is kept unchanged. What happens to the average speed of the hydrogen molecules?
-
In Exercises 516, find the focus and directrix of the parabola with the given equation. Then graph the parabola. y 2 = 4x
-
Write the balanced molecular and net ionic equations for each of the following neutralization reactions: (a) Aqueous acetic acid is neutralized by aqueous barium hydroxide. (b) Solid chromium(III)...
-
Write balanced molecular and net ionic equations for the following reactions, and identify the gas formed in each:
-
Magnesium carbonate, magnesium oxide, and magnesium hydroxide are all white solids that react with acidic solutions. (a) Write a balanced molecular equation and a net ionic equation for the reaction...
-
A 7 5 kg cliff - diver is falls from a height through the air head, diving towards the water. The drag coefficient for the diver is 0 . 8 3 and the area of the descending diver is 0 . 2 1 m ^ 2 ....
-
In the automobile industry, the dimensionless drag coefficient and the area of the vehicle are often combined into one variable - the drag area whereby the drag area is the product of the...
-
When you drop 10 pebbles into the well, you record the times for hearing the splash as 2.94 s, 3.11 s, 3.12 s, 2.97 s, 3.12 s, 2.97 s, 3.06 s, 3.21 s, 3.37 s, and 3.53 s. (a) Find the average time...

Study smarter with the SolutionInn App


