Tell whether the following compounds are ortho-, meta-, orpara-di-substituted: (a) CI. CH3 (b) NO2 (c) SO3H Br
Question:
Tell whether the following compounds are ortho-, meta-, orpara-di-substituted:
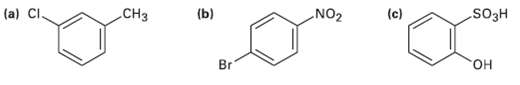
Transcribed Image Text:
(a) CI. CH3 (b) NO2 (c) SO3H Br он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
An ortho disubstituted benzene has two substituents in a 12 ...View the full answer

Answered By

Patrick Busaka
I am a result oriented and motivated person with passion for challenges because they provide me an opportunity to grow professionally.
5.00+
38+ Reviews
58+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Tell whether the following compound is chiral. trans- I,2-dimethylcyclopropane
-
The following compounds are only slightly soluble in water, but one of them is very soluble in a dilute aqueous solution of sodium hydroxide. The other is still only slightly soluble. (a) Explain...
-
The following compounds are listed in increasing order of acidity. In each case, the most acidic proton is shown in red. (a) Show the structure of the conjugate base of each acid, including any...
-
A ping pong ball is drawn at random from an urn consisting of balls numbered 4 through 9. A player wins $1.5 if the number on the ball is odd and loses $1.5 if the number is even. Let x be the amount...
-
If you were advising a North Dakota company about its selection process, would you advise it to relax its selection criteria during the oil boom? Why or why not?
-
Amber and Travis are considering careers in business and have hopes of becoming managers someday. Both hold part-time jobs and have seen a number of managers at their work. Amber was even selected to...
-
Show that the inequality on page 304 leads to the following \((1-\alpha) 100 \%\) confidence limits: \[\frac{x+\frac{1}{2} z_{\alpha / 2}^{2} \pm z_{\alpha / 2} \sqrt{\frac{x(n-x)}{n}+\frac{1}{4}...
-
1. What type of contact is between the Ridpath Sandstone and the Kavalier Shale? How did you determine this? (Refer to the Geologic Time Scale, which is Figure 1 in the Geologic Dating lab, to help...
-
Company had $ 8 5 , 3 8 0 of accounts payable on september 1 and $ 7 6 , 5 6 0 on september 3 0 . During September , the company paid total cash of $ 7 9 , 6 5 0 on accounts payable. Determine the...
-
The complexity and uniqueness of the federal government make it difficult to conduct a meaningful analysis of its financial condition. However, conducting a financial statement analysis does provide...
-
Amines are converted into alkenes by a two-step process called the Hofmann elimination. SN2 reaction of the amine with an excess of CH3I in the first step yields an intermediate that undergoes E2...
-
Give IUPAC names for the followingcompounds: CH NH2 (a) Cl Br (b) (c) CH2CH2CHCH3 Br (f) CH (e) (d) CI. CH H CH-CH O2N" NO2 "C
-
Compute the products in Exercises 1-2 using (a) the definition, as in Example 1, and (b) the row-vector rule for computing Ax. If a product is undefined, explain why. 1. 2. 327
-
Review your local newspapers or online sources for current discount, federal funds, and prime rates, plus average mortgage interest rates and report on what you found. This means elaborate on what...
-
What is investor in common stock? and Discuss at least three specific financial ratios that creditors or investors would be most interested in when analyzing financial statements, and why?
-
Calculate (and document) suitable ratios with respect to of a) liquidity, b) solvency, c) profitability and d) efficiency BRIEFLY (maximum of 400 words) use these ratios to respond to the following 3...
-
A Treasury strip with 10-year maturity and $100 par value has a current price of $55.84. Suppose that the interest rate drops over the next year by 1 percent. What is the holding period return on...
-
The portfolio steering committee is considering selecting Project A. The committee expects the project will produce a one-time benefit of 2.5 Million Dollars three years from now. The interest rate...
-
Brenda Johnson is considering purchasing a new car for business purposes. She has narrowed her choice to three cars; the cost details are given below: Brenda estimates she will travel about 4,000...
-
A genetically engineered strain of Escherichia coli (E. coli) is used to synthesize human insulin for people suffering from type I diabetes mellitus. In the following simplified reaction scheme,...
-
Explain how you might deduce the equilibrium constant for a reaction in which you know the initial concentrations of the reactants and products and the equilibrium concentration of only one reactant...
-
Propose a mechanism for the base-catalyzed epimerization of erythrose to a mixture of erythrose and threose.
-
Show how another enediol rearrangement can move the carbonyl group from C2 in fructose to C3.
-
Show how another enediol rearrangement can move the carbonyl group from C2 in fructose to C3. Discuss.
-
Johnny's Lawn Service camed $200,000 of service revenues for 2020. $190,000 was collected in cash and $10,000 remains in Accounts Receivable as of 12/31/20. S155,000 of expense was incurred during...
-
Company BW has $500,000 loan outstanding. The annual loan interest rate is 6% with monthly compounding (meaning BW is making monthly payments). Find the annual after-tax cost of debt for the loan.
-
The hierarchical system of classification is intended to be a map of evolution. Explain this statement. What is implied about species that occur on the same branch of a phylogenetic tree?

Study smarter with the SolutionInn App


