Question: Terminal alkynes react with Br2 and water to yield bromo ketones. For example: Propose a mechanism for the reaction. To what reaction of alkenes is
Terminal alkynes react with Br2 and water to yield bromo ketones. For example: Propose a mechanism for the reaction. To what reaction of alkenes is the processanalogous?
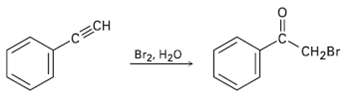
-CECH Br2, H20 CH2Br
Step by Step Solution
3.32 Rating (161 Votes )
There are 3 Steps involved in it
This reaction mechanism is similar to the mechan... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-O-S (45).docx
120 KBs Word File


