The (a) mass spectrum and the (b) infrared spectrum of an unknown hydrocarbon are shown. Propose as
Question:
The (a) mass spectrum and the (b) infrared spectrum of an unknown hydrocarbon are shown. Propose as many structures as youcan.
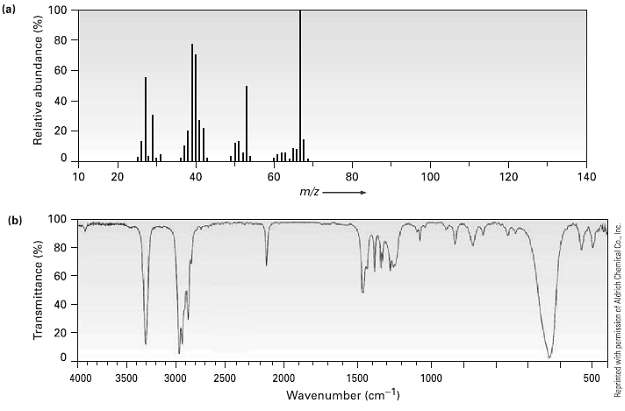
Transcribed Image Text:
(a) 100 80 60 40 20 10 20 40 60 80 100 120 140 m/z (b) 60 40 20 - 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-1) Transmittance (%) Relative abundance (%) Reprinted with permissicn et Aldrich Chemical Co. Inc.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The peak of maximum intensity base peak in the mass spectrum occurs at mz 67 This ...View the full answer

Answered By

Seema kuldeep
although I don't have an experience of teaching in a particular institute, previously I was an expert on Chegg and I have used to teach my batch mates and also my juniors.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The mass spectrum and infrared spectrum of an unknown compound are shown in Figures 13.27 and 13.28, respectively. Identify the compound. Figure 13.27 The mass spectrum for Problem 28. 100 E 80 3 60...
-
An unknown compound gives a mass spectrum with a weak molecular ion at m/z 113 and a prominent ion at m z 68. Its NMR and IR spectra are shown here. Determine the structure, and show how it is...
-
An unknown, foul-smelling hydrocarbon gives the mass spectrum and infrared spectrum shown. (a) Use the mass spectrum to propose a molecular formula. How many elements of unsaturation are there? (b)...
-
Reply as to whether you believe the following statements are correct (C) or incorrect (I) concerning PPS sampling. a. The size of a PPS sample is not based on the estimated variation of audited...
-
An article on price wars by two McKinsey consultants makes the following argument. That the (tit-for-tat) strategy is fraught with risk cannot be overemphasized. Your competitor may take an...
-
For a random sample of 60 overweight men, the mean of the number of pounds that they were overweight was 30. The standard deviation of the population is 4.2 pounds. a. Find the best point estimate of...
-
Describe how you think history is repeating itself in todays healthcare system.
-
Residents of Mill River have fond memories of ice skating at a local park. An artist has captured the experience in a drawing and is hoping to reproduce it and sell framed copies to current and...
-
Required 1. Required 2 Required 3 s below. Stone needs to accumulate sufficient funds to pay a $490,000 debt that comes duelon December 31, 2029. The company will accumulate the funds by making five...
-
The actuary for the pension plan of Gustafson Inc. calculated the following net gains and losses Incurred during the Year (Gain) or Loss 2020 $300,000 2021 480,000 2022 (210,000) 2023 (290,000) Other...
-
Carvone (Problem 12.39) has an intense infrared absorption at 1690 cm1. What kind of ketone does carvone contain?
-
The (a) mass spectrum arid the (b) infrared spectrum of another unknown hydrocarbon is shown. Propose as many structures as youcan. (a) 100 80 60 40 20 20 60 80 100 120 140 10 40 m/z 100 (b) * 80 60...
-
Let x be a discrete random variable that possesses a binomial distribution. Using the binomial formula, find the following probabilities. a. P(x = 0) for n = 5 and p = .05 b. P(x = 4) for n = 7 and p...
-
Identify at least five contemporary technology trends and/or issues relevant to your anticipated career path, and write a business memo on the technology trends and a project idea. Be realistic in...
-
Identify which technological tools you plan to use for your organization and explain why?
-
INCOME Everyday Flowers and Workshops Sales of Product Income Special Occasions Total Income COST OF GOODS SOLD Flower Purchases and Supplies Supplies and materials - COS Total Cost of Goods Sold...
-
First, let's see what we can learn about retirement needs from the TVM calculations we know how to do. You expect to need $75,000 a year in retirement and you expect your retirement to last 35 years....
-
Noemi strings together x green beads at $0.75 each with 3 blue beads at $0.30 each. She makes a bracelet that averages $0.60 per bead. How many green beads does Noemi use? green beads
-
Can your diet make you happier? A researcher wants to know if the food you eat has an effect on your happiness. A random sample of 41 participants was asked to complete a survey on happiness and a...
-
1) Predict the organicproduct formed when BzCl reacts with cyclohexanol. BzCl = benzoylchloride. 2) Provide the majororganic product of the reaction below. 3) Draw the structureof the product formed...
-
The Safe Drinking Water Act (SDWA) sets a limit for mercurya toxin to the central nervous systemat 0.0020 ppm by mass. Water suppliers must periodically test their water to ensure that mercury levels...
-
Dimethylamine, (CH3)2NH has a molecular weight of 45 and a boiling point of 7.4 C. Trimethylamine, (CH3)3N has a higher molecular weight (59) but a lower boiling point (3.5 C). Explain this apparent...
-
Predict which member of each pair will be more acidic. Explain your answers. (a) Methanol or tert-butyl alcohol (b) 2-chloropropan-1-ol or 3-chloropropan-1-ol (c) 2-chloroethanol or...
-
Without looking them up, rank the following compounds in decreasing order of acidity. These examples represent large classes of compounds that differ widely in acidity. water, ethanol,...
-
Given the function. f(x) = ln (2x+3x) Compute f'(x). Enter your answer rounded to the nearest tenth
-
A motor-cycle manufacturing company desires a profit of $600,000. The fixed costs are $800,000. The unit selling price of the motor-cycle is $750 and the variable cost per unit is $680. How many...
-
3. ZR Corporation's stock has a beta coefficient equal to 0.8 and a required rate of return equal to 11 percent. If the expected rate of return on the market is 12.5 percent, what is the risk-free...

Study smarter with the SolutionInn App


