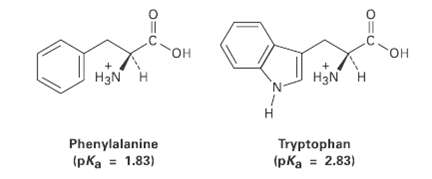
The amino acid phenylalanine has pKa = 1.83, and tryptophan has pKa = 2.83. Which is the
Question:
The amino acid phenylalanine has pKa = 1.83, and tryptophan has pKa = 2.83. Which is the strongeracid?
Transcribed Image Text:
он он OH H3N H H3N H н Tryptophan Phenylalanine (pka = 1.83) (pka = 2.83)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
Recall from Section 28 that a stro...View the full answer

Answered By

Rashul Chutani
I have been associated with the area of Computer Science for long. At my university, I have taught students various Computer Science Courses like Data Structures, Algorithms, Theory of Computation, Digital Logic, System Design, and Machine Learning. I also write answers to questions posted by students in the area of and around Computer Science.
I am highly fortunate to receive great feedback on my teaching skills that keeps me motivated. Once a student sent me an email stating that I had explained to him a concept better than his professor did.
I believe in the fact that "Teaching is the best way to learn". I am highly fascinated by the way technology nowadays is solving real-world problems and try to contribute my bit to the same.
Besides tutoring, I am a researcher at the Indian Institute of Technology. My present works are in the area of Text Summarization and Signal and Systems.
Some of my achievements include clearing JEE Advanced with an All India Rank of 306 out of 1.5 million contesting candidates and being the Department Ranker 1 at my University in the Department of Computer Science and Engineering.
I look forward to providing the best Tutoring Experience I can, to the student I teach.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Acid HA has pKa = 20; acid HB has pKa = 10. (a) Which is the stronger acid? (b) Will an acid-base reaction with an equilibrium lying to the right take place if Na+A- s added to HB? Explain your...
-
The acid HA has pKa = 7.00. (a) Which is the principal species, HA or A-, at pH 6.00? (b) Which is the principal species at pH 8.00? (c) What is the quotient [A-]/[HA] at pH 7.00? at pH 6.00?
-
The amino acid cysteine has the structure shown: (a) A second sulfur-containing amino acid called cystine (C6H12N2O4S2) is formed when cysteine undergoes biological oxidation. Suggest a reasonable...
-
Why is it so difficult to assess whether a firm is properly valued?
-
Cisco, a calendar year taxpayer who is age 63, owns a residence in which he has lived for 21 years. The residence is destroyed by fire on August 8, 2016. The adjusted basis is $190,000, and the fair...
-
Kasson Company has budgeted units to be produced for the next three months as follows: April May June budgeted units to be produced 46,000 73,000 ?????? Kasson Company used the following information...
-
Show that for the Ho-Lee model the (risk-neutral) standard deviation of the one-period rate is exactly \(b_{k} / 2\).
-
Define experimental condition, experimental treatment, and experimental group. How are these related to the implementation of a valid manipulation?
-
Please choose a green brand and develop and answer a report answering the following questions: 1) How do you see the green "sensibility" in their ads - analyze the message 2) Analyze the brand...
-
FOR YEAR5 sourcing has been just another word for procurement a financially material but strategically peripheral corporate function Now globalization aided by rapid technology innovation is changing...
-
Nitric acid (HNO3) reacts with ammonia (NH3) to yield ammonium nitrate. Write the reaction, and identify the acid, the base, the conjugate acid product, and the conjugate base product.
-
Amide ion, H 2 N , is a stronger base then hydroxide ion, HO . Which is the stronger acid, NH 3 or H 2 O? Explain.
-
How might you design a transferable discharge permit system for solid waste? For phasing out of use a certain type of plastic? For phasing in a program for using recycled newsprint in newspapers?
-
Name several operating systems that run LANs today. Do not include different versions of a single operating system. For each operating system, list its name, the platform or network it operates on,...
-
What does it mean by product differentiation? How can enterprises market their products different from similar products offered by competitors? What roles do you think information technology and...
-
What are the challenges and opportunities associated with harnessing microbial biodegradation pathways for environmental remediation and sustainable bioprocessing initiatives?
-
How do halogens work? Give examples.
-
Unix Operating Systems and User Interface ----------------------------------------------------------------------------------------------------------------- Q. Mobile Operating System Research and...
-
How can the prosecution establish the corpus delicti in a murder case when the body of the victim cannot be found?
-
During the year land was revalued and the surplus reported as Revaluation surplus; and an asset costing 80,000, written down to 38,000, was sold for 40,000. Identify the cost of any non-current...
-
a. Using only the steam tables, compute the fugacity of steam at 400C and 2 MPa, and at 400C and 50 MPa. b. Compute the fugacity of steam at 400C and 2 MPa using the principle of corresponding...
-
Name the following compounds: a) c) H C CH3 b) d)
-
Draw structures for these compounds: (a) 1, 1-Dimethylcylohexane (b) Ethylcyclopropane
-
Name these compounds: 22 b)
-
6x+17x-3 Evaluate the following limit: lim xo 5x2+14x-3
-
Find all the critical numbers of y=x-2x-3, then determine the local minimum and maximum points by using a graph.
-
At September 3 0 , the end of Beijing Companys third quarter, the following stockholdersequity accounts are reported. Common stock, $ 1 0 par value $ 4 8 0 , 0 0 0 Paid - in capital in excess of par...

Study smarter with the SolutionInn App


