The cycle in Figure represents the operation of a gasoline internal combustion engine. Volume V3 = 4.00V1.
Question:
The cycle in Figure represents the operation of a gasoline internal combustion engine. Volume V3 = 4.00V1. Assume the gasoline - air intake mixture is an ideal gas with γ = 1.30 what are the ratios
(a) T2/T1
(b) T3/T1,
(c) T4/T1
(d) p3/p1,
(e) p4/p1?
(f) What is the engine efficiency?
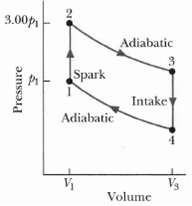
3.00p Adiabatic Spark Intake Adiabatic V3 Volume Pressure
Step by Step Answer:
a The pressure at 2 is p2 300p as given in the problem statement The volume is V VnRTp The temperatu...View the full answer



Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Related Video
An air pump is a device used to inflate or deflate objects by moving air in or out of them. There are many different types of air pumps, ranging from manual hand pumps to electrically powered pumps, and they are used for a variety of purposes. John had trouble blowing up many balloons without a balloon pump and experienced pain. He solved this problem by using an empty plastic bottle with a rectangular-shaped hole in the cap, a cut piece of balloon stretched over it, and a small hole in the bottle. By pressing and releasing the bottle, he created an air pump to inflate the balloons without any discomfort. Air pumps utilize fluid mechanics and thermodynamics principles to increase air pressure inside an object being inflated, such as a bicycle tire. The ideal gas law is used to calculate the pressure inside the tire. As air pressure increases, the object becomes more rigid and can support more weight. The efficiency of the pump is determined by how much of the work done is converted into useful work, such as inflating the tire, and how much is lost as heat. Understanding these principles is important to effectively use and optimize the use of air pumps.
Students also viewed these Thermodynamics questions
-
An ideal gas with the adiabatic exponent goes through a cycle (Fig. 2.3) within which the absolute temperature varies -fold. Find the efficiency of this cycle.
-
An ideal gas with the adiabatic exponent y goes through a process p = Po- aV, where Po and a are positive constants, and V is the volume. At what volume will the gas entropy have the maximum value?
-
An ideal gas with k = 1.4 is flowing through a nozzle such that the Mach number is 2.4 where the flow area is 25 cm2. Assuming the flow to be isentropic, determine the flow area at the location where...
-
Two kilograms of water, initially saturated liquid at 10 kPa, are heated to saturated vapor while the pressure is maintained constant. Determine the work and the heat transfer for the process, each...
-
Why is it that the greater the number of firms is in a monopolistically competitive industry, the lower the price is, but the higher the average cost of each firm is for a given level of output?
-
Yahoo! has expanded its business by establishing portals in numerous countries, including Argentina, Australia, China, Germany, Ireland, Japan, and the U.K. It has cash outflows associated with the...
-
Calculate the mean for each of the following sets of data: a. 2, 2, 3, 5 b. 3, 5, 5, 7, 7, 8, 8, 11 c. 9, 6, 7, 5, 8, 7, 8, 9, 10, 8, 7 d. 11, 13, 14, 15, 16, 16, 16, 17, 18, 20, 22, 25 e. 2.57,...
-
Air travel on Mountain Airlines for the past 18 weeks was: a. Explain why an averaging technique would not be appropriate for forecasting. b. Use an appropriate technique to develop a forecast for...
-
How does Lean Management foster a culture of continuous improvement and waste reduction within organizational processes ? Explain
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
In the first stage of a two-stage Carnot engine, energy is absorbed as heat Q1 at temperature T1, work W1 is done, and energy is expelled as heat Q2 at a lower temperature T2. The second stage...
-
The electric motor of a heat pump transfers energy as heat from the outdoors, which is at 5.0oC, to a room that is at 17oC.If the heat pump were a Carnot heat pump (a Carnot engine working in...
-
In 2014, Wild Corporation reported a net loss of $70,000. Wild's only net income adjustments were depreciation expense $81,000, and increase in accounts receivable $8,100. Compute Wild's net cash...
-
The crankshaft of a four-cylinder in-line air compressor engine rotating with a speed of oo is shown in the figure. For each cylinder, the crank radius is 25 mm, the connecting rod length is 90 mm...
-
Assume y= f(x1, x2) = min(ax, bx2) If bx1 < ax2 what is the marginal product of the first input? What is the marginal product of the second input? What is the returns to scale? 2. What is TRS if y...
-
J&J stock price is currently trading at $100. Over each of the next two three-month periods it is expected to go up by 8% or down by 7%. The risk-free interest rate is 5% per annum with continuous...
-
Use TABLE ONE to show the difference it makes, in the long run, if a particular level of output relies more on increased capital stock than on a higher level of technology. The table shows the lower...
-
On one k-y graph, show the economy a) in the original equilibrium in year zero, b) in the original equilibrium in year ten. c) in year three d) in the new equilibrium in year ten, with reduced g but...
-
Macquarie Manufacturing Ltd prepared the following planned production data for the forthcoming year ending 30 June 2019. Required (a) Prepare a table showing the predetermined factory overhead rate...
-
Use Stokes' Theorem to evaluate f(y+sin x) dx+(z+cos y) dy+rdz, where C is the rve r(t) = (sint, cost, sin 2t), t = [0, 2].
-
A small pickup truck that has a camper shell slowly coasts toward a red light with negligible friction. Two dogs in the back of the truck are moving and making various inelastic collisions with each...
-
Modern vacuum pumps make it easy to attain pressures of the order of 10-13 atm in the laboratory. (a) At a pressure of 9.00 X 10-14 atm and an ordinary temperature of 300.0 K, how many molecules are...
-
The Lagoon Nebula (Fig. 18.28) is a cloud of hydrogen gas located 3900 light-years from the earth. The cloud is about 45 light years in diameter and glows because of its high temperature of 7500 K....
-
In a gas at standard conditions, what is the length of the side of a cube that contains a number of molecules equal to the population of the earth (about 6 X 109 people)?
-
Georgias employee will match her contributions into a retirement plan up to 5% of her 40,000 annual salary. in other words, the employee will put $1 into her retirement plan for every $1 Georgia puts...
-
Stock in Country Road Industries has a beta of 1.09. The market risk premium is 7.5 percent, and T-bills are currently yielding 3.5 percent. The company's most recent dividend was $1.7 per share, and...
-
Festival Dancing and Fitness In this activity, you will be provided with a review on the implication of dancing activity to your fitness by way of determining your range of Target Heart Rate. Let us...

Study smarter with the SolutionInn App


