The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure
Question:
The energy level diagram for the MOs of CH2 = CH2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule.
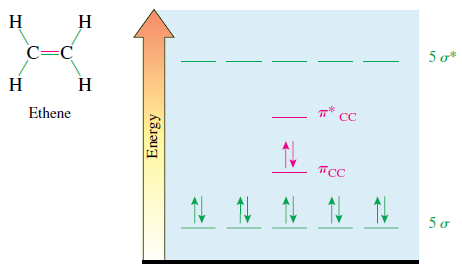
Transcribed Image Text:
н н C=C 5 o* н Н т* сс Ethene Псс 5 o Energy
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (18 reviews)
The lowest energy excited st...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show energy level diagram for the MOs of these compounds? a) H-C=N: b) H 0: C CH3 ) CHNH,
-
Show an energy level diagram for the MOs for He 2 and show how the electrons would be arranged in these MOs.
-
Figure 29.7 is an energy level diagram for 208Tl. What are the energies of the photons emitted for the six transitions shown? Figure 29.7 492 keV 472 keV 327 keV 40 keV
-
All adiabatic temperature changes occur as a result of a. changes in the absolute humidity of the air. b. expansion or compression of the air. c. changes is specific humidity d. the addition or...
-
The only variable overhead cost at Jackson Sporting Club is electricity. Does an unfavorable variable overhead spending variance imply that the club paid more than the anticipation per kilowatt hour?
-
On February 28, 2012, Stu & Dent, LLP completed the audit of Shylo Ranch, Inc. (a nonpublic company) for the year ended December 31, 2011. A recent fire destroyed the accounting records concerning...
-
Identify three business rules for an ATM system.
-
Valuing Bonds The Marigold Corporation has two different bonds currently outstanding. Bond M has a face value of $20,000 and matures in 20 years. The bond makes no payments for the first six years,...
-
Primare Corporation has provided the following data concerning last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor $ 32,000 $ 4,860 $...
-
The number of calories per candy bar for a random sample of standard-size candy bars is shown below. Estimate the mean number of calories per candy bar with 98% confidence. Assume that all variables...
-
Show energy level diagrams for the MOS of these compounds: a) H-C=C-CH3 b) CH0H c) CH3-C-H
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
To get $300 after 4 years at 3% compounded daily In problem, find the principal needed now to get each amount; that is, find the present value.
-
A bag holds 10 black jellybeans, 12 green ones, 3 orange ones, and 20 blue ones. If you reach in and grab one randomly, what is the probability of picking sequentially (a) a black one, \((b)\) an...
-
Particles in a closed container move around and collide with one another and with the walls of the container. There are \(5.00 \times 10^{13}\) basic states possible for this system, and the...
-
When two standard dice are thrown, what is the probability that the sum of the dots on the two top faces will be 7 ?
-
With a standard deck of 52 playing cards, what is the probability of randomly picking (a) any card numbered 8 and \((b)\) any king, queen, jack, or ace?
-
Water leaks out of a small hole in the side of a bucket. The hole is a distance \(d\) below the surface of the water, and the cross section of the hole is much smaller than the diameter of the...
-
As incongruous as it seems, the Hangover movie franchise is a hotbed of intellectual property issues. Last summer, Warner Brothers settled a lawsuit brought by the tattoo artist who did Mike Tyson's...
-
Wholesalers Ltd. deals in the sale of foodstuffs to retailers. Owing to economic depression, the firm intends to relax its credit policy to boost productivity and sales. The firms current credit...
-
Find the Maclaurin series of the function f(x) = e x and its radius of convergence.
-
How many H H eclipsing interactions would be present if cyclopentane were planar? Assuming an energy cost of 4.0kJ/mol for each eclipsing interaction, how much torsional strain would planar...
-
Two conformations of cis-1, 3-dimethylcyclobutanc is shown. What is the difference between them, and which do you think is likely to be more stable? (b) (a)
-
Draw two different chair conformations of cyclohexanol (hydroxycyclohexane), showing all hydrogen atoms. Identify each position as axial or equatorial.
-
1 Mandatory requirement: Students are required to complete Budget on the provided Excel 2 3 4 5 5 B spreadsheet template. (For the purpose of Budget calculation in the assessment, ignore all GST...
-
A company has Rs 4 per year carrying cost on each unit of inventory, an annual usage of 50,000 units and an ordering cost of Rs 100 per order. Calculate the economic order quantity. What shall be the...
-
The company decided to make some changes to its operations based on the actual results and the variance analysis. The plan is to maintain the selling price of the product at RM100 per unit and the...

Study smarter with the SolutionInn App


