Show energy level diagrams for the MOS of these compounds: a) H-C=C-CH3 b) CH0H c) CH3-C-H
Question:
Show energy level diagrams for the MOS of these compounds:
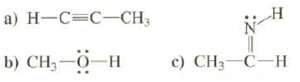
Transcribed Image Text:
a) H-C=C-CH3 b) CH—0—H c) CH3-C-H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a HCCCH3 E 1 c H CH3CH ...View the full answer

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show energy level diagram for the MOs of these compounds? a) H-C=N: b) H 0: C CH3 ) CHNH,
-
The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule. C=C 5 o* * Ethene 5 o Energy
-
Draw molecular orbital energy level diagrams for O2, O2-, and O22-.
-
Cindy purchased a building for $1,000,000. How should she deal with the expense on her tax return? a) She must deduct the $1,000,000 in the year of purchase. b) She must not deduct anything and wait...
-
Explain the meaning of the variable overhead efficiency variance?
-
The following auditor's report was drafted by a staff accountant of Turner & Turner, CPAs, at the completion of the audit of the financial statements of Lyon Computers, Inc. (a nonpublic company) for...
-
Identify three objects for an ATM system.
-
Refer to the data provided in E4A. In E4A. In chronological order, the inventory, purchases, and sales of a single product for a recent month are as follows. 1. Using the perpetual inventory system,...
-
need help to mark my working and answer and provide explanation if wrong Image transcription text A power plant operates with a high-pressure steam of 50 bar and has a boiler exit temperature of 600...
-
The number of calories per candy bar for a random sample of standard-size candy bars is shown below. Estimate the mean number of calories per candy bar with 98% confidence. Assume that all variables...
-
Explain why one of these carbocations is much more stable than the other: + CH-CH3 CH CH
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
Exercise 6 Cell Phone Data From Exercise 6 Claim: Fewer than 95% of adults have a cell phone. In a Marist poll of 1128 adults, 87% said that they have a cell phone. In refer to the exercise...
-
Joey offers to sell Melissa the latest version of a popular gaming system for only $25 cash, although in the store it generally costs over $200. The deal seems too good to be true, and Melissa...
-
Carrie is addicted to heroin and will do anything to feed her addiction. She decides to take her husbands Rolex watch and sell it at the local pawnshop for cash. The Rolex watch was purchased with...
-
False pretenses is shorthand for the crime of obtaining property by deception. It is distinct from larceny because of the deception and/or false representation involved. Forgery is the false making...
-
When a piece of cork floats in water, three-quarters of its volume is above the water surface. How does the mass density of cork compare with the mass density of water?
-
A dam of horizontal length \(\ell\) holds water of mass density \(ho\) to a height \(h\). What is the magnitude of the force exerted by the water on the dam?
-
Electronic Arts, Inc., a video company, had developed football and college video games. However, many of the players in the video games seemed to be images and likenesses of actual college athletes...
-
Using Gauss-Jordan elimination, invert this matrix ONLY 0 0 0 0 1
-
Find the sum of the series correct to three decimal places. (By definition, 0! = 1.) 00 n=0 (-1)" n!
-
Names the following substances, including the cis-or trans-prefix (red-brown =Br): (b) (a)
-
Each H H eclipsing interaction in ethane costs about 4.0kJ/mol. How many such interactions are present in cyclopropane? What fraction of the overall 115 kJ/mol (27.5kcal/mol) strain energy of...
-
Cis-l, 2-Dimethylcyclopropane has more strain than trans-1, 2-dimethylcyclo- propane. How can you account for this difference? Which of the two compounds is more stable?
-
The community nursing service team provides nursing care to patients in the community, Discuss?
-
Why are angiosperms so diverse (when compared with non-flowering lineages of plants)? How expensive flowers are to produce for the plant (they cost lots of ATP to make); what benefit do they provide...
-
Martinez Company's relevant range of production is 7,500 units to 12,500 units. When it produces and sells 10,000 units, its average costs per unit are as follows: Average Cost Per Unit Direct labor...

Study smarter with the SolutionInn App


