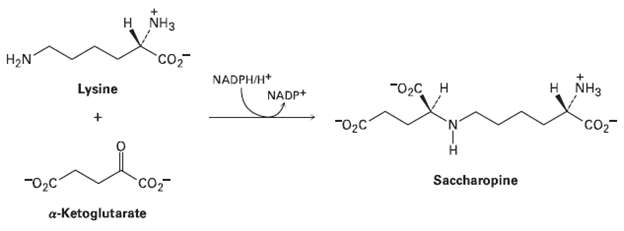
The first step in the biological degradation of lysine is reductive amination with ?-ketoglutarate to give saccharopine.
Question:
The first step in the biological degradation of lysine is reductive amination with ?-ketoglutarate to give saccharopine. Nicotinamide adenine dinucleotide phosphate (NADPH), a relative of NADH, is the reducing agent. Show the mechanism.
Transcribed Image Text:
H NH3 Co H2N NADPH/H+ NADP+ Lysine H NH3 "02с н 02C 'N' CO2 Saccharopine Co a-Ketoglutarate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 47% (17 reviews)
00 0 0C HN CO O CO H2O Nucleophilic ad...View the full answer

Answered By

ALBANUS MUTUKU
If you are looking for exceptional academic and non-academic work feel free to consider my expertise and you will not regret. I have enough experience working in the freelancing industry hence the unmistakable quality service delivery
4.70+
178+ Reviews
335+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The first step in the revenue recognition process is determining if a contract is in place between the seller and the customer. A contract is an agreement between two or more parties that creates...
-
The first step in the controlling process involves measuring performance. If you were John Thain, how would you measure the performance of the financial planners? Be specific.
-
The first step in eq. 10.26 really involves two reactions, addition of ammonia to the carbonyl carbon to form an ammonium alkoxide followed by a proton transfer from the nitrogen to the alkoxide...
-
List four components and four guidelines that the J. Crew mission statement fails to exhibit. Write a new and improved mission for J. Crew.
-
Select a piece of business mail that you received at work or at home. Analyze its appearance. What nonverbal messages does this piece send? Are these messages consistent with the content of the...
-
a. Given Pmax = 14 mW for each diode at Fig. 2.167, determine the maximum current rating of each diode (using the approximate equivalent model). b. Determine/max for Vmax for Vimax = 160 V. c....
-
Superheated steam at \(580^{\circ} \mathrm{F}\) and \(500 \mathrm{psia}\) is expanded across a turbine, as shown in Figure 10.31, to \(540^{\circ} \mathrm{F}\) and 400 psia. \(0.9 \mathrm{~kW}\) of...
-
Leach Company borrowed $80,000 cash by issuing a note payable on June 1, Year 1. The note had an 8 percent annual rate of interest and a one-year term to maturity. Required a. What amount of interest...
-
Major medical complexes and their service providers continue to move toward advanced health informatics - acquiring, managing, and using information to provide better healthcare. A new analyzer for...
-
In the Bombadier Company, Division A has a product that can be sold either to outside customers or to Division B. Information about these divisions is given below: Case 1 Case 2 Division A: Capacity...
-
The first step in the biological degradation of histidine is formation of a 4-methylidcneirnidazol-5-one (MIO) by Cyclization of a segment of the pep- tide chain in the histidine ammonia lyase...
-
Carnauba wax, used in floor and furniture polishes, contains an ester of a C32 straight-chain alcohol with a C20 straight-chain carboxylic acid. Draw its structure.
-
A small price-taking nation imports a good that it could not possibly produce itself at a finite price. Can you describe a plausible condition under which that nation would benefit from an import...
-
A colleague working with you in the same design office has completed a seismic analysis and design for a 5-story reinforced concrete building that satisfies all the checks according to Eurocode 8....
-
Chris is going to make monogrammed polar fleece scarves and sell them for $10.00 each. His fixed costs of $860 include a sewing machine and a cell phone. In order to produce 30 scarves Chris will...
-
Following is information regarding inventory transactions sold by SouthCo. The transactions occured in the order shown. Date Transaction Unit Cost $4.00 4.30 22/Feb 11/Apr 3. Sales (at $12.50 per...
-
9. As cited in an earlier example in class, an automatic shifting machine inserts mixed vegetables into a plastic bag. Experience revealed some packages were underweight and some overweight, but most...
-
Given is the following NFA with three states: 1, 2, and 3. Start 2 Final 3 1 QUESTION 8 Given is the following NFA with three states: 1, 2, and 3. In the context of converting this NFA to DFA...
-
Mario and Claudia deposit $300 into their joint account at the end of each quarter. If their account earns 7 percent/ year/month (7 percent per year compounded monthly), how long will it take them to...
-
(a) Find the equation of the tangent line to f(x) = x 3 at the point where x = 2. (b) Graph the tangent line and the function on the same axes. If the tangent line is used to estimate values of the...
-
For each strong base solution, determine [OH ], [H 3 O + ], pH, and pOH. a. 0.15 M NaOH c. 4.8 x 10-4 M Sr(OH)2 b. 1.5 x 10 M Ca(OH)2 d. 8.7 x 10 5 M KOH
-
Describe the expected 1H NMR spectrum of a. b. CH3-C¡C - H ,
-
The max for cis-1,2-diphenylethene is at shorter wavelength (280 nm) than for trans-1,2 diphenylethene (295 nm). Suggest an explanation.
-
A sample of methylcyclohexane is suspected of being contaminated with toluene, from which it had been prepared by hydrogenation. At 261 nm, toluene has a molar absorptivity = 224, whereas...
-
Image transcription text 1. A cylindrical specimen of cold-worked copper (see Fig. below) has experienced a ductility of 25%EL. If its cold worked radius is 10 mm, what was its radius before...
-
Question 2 As a project manager, you need to synthesize information about a problem to stakeholders. What should you do first in your synthesizing process? Describe Briefly.
-
Please help me as much as you can!! I will take all your effort to solve this problem and will give you a good rate!!! Please show all the calculations in detail! Also, please do not copied and...

Study smarter with the SolutionInn App


