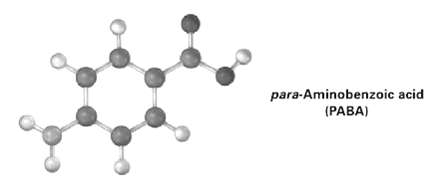
The following molecular model is a representation of para-aminobenzoic acid (PABA), the active ingredient in many sunscreens.
Question:
The following molecular model is a representation of para-aminobenzoic acid (PABA), the active ingredient in many sunscreens. Indicate the positions of the multiple bonds. And draw a skeletal structure (gray = C, red = O, blue = N, ivory =H).
Transcribed Image Text:
para-Aminobenzoic acid (PABA)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
HN Vis...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following ball-and-stick molecular model is a representation of thalidomide, a drug that causes birth defects when taken by expectant mothers (red = 0, gray = C. blue = N, ivory = H). (a) What is...
-
The following model is a representation of asparatame, C14H18N2O5, known commercially as NutraSweet. Only the connection between atoms is shown; multiple bonds are not indicated. Complete the...
-
The following molecular model is that o a carbocation. Draw two resonance structures for the carbocation, indicating the positions of the doublebonds.
-
How can a layer fold and boudinage (extend) at the same time?
-
Osprey Corporation, an accrual basis taxpayer, had taxable income for 2016 and paid $40,000 on its estimated state income tax for the year. During 2016, the company received a $4,000 refund upon...
-
When pseudoionone is treated with BF3 in acetic acid, ring closure takes place and α-and β-ionone are produced. This is the next step in the vitamin A synthesis. (a) Write...
-
The gas entering a rocket nozzle has a stagnation pressure of \(1500 \mathrm{kPa}\) and a stagnation temperature of \(3000^{\circ} \mathrm{C}\). The rocket is traveling in the still Standard...
-
Use the ending balances from Problem 10-24 to prepare a balance sheet for CASH for 2013.
-
Criminal possession of a weapon in the second degree. A person is guilty of criminal possession of a weapon in the second degree when: (1) with intent to use the same unlawfully against another, such...
-
1. Sharps Sandwich Shop has two conflicting quality issues speed and freshness. The premade sandwich system enhances speed; however, it has the potential of affecting freshness. What type of system...
-
Propose skeletal structures for compounds that satisfy the following molecular formulas. There is more than one possibility in each case. (a) C5H12 (b) C2H7N (c) C3H6O (d) C4H9Cl
-
Convert each of the following molecular models into skeletal structure, and give the formula of each. Only the connections between atoms are shown; multiple bonds are not indicated (gray = C, red ?...
-
Which of the following shows the correct balance of Deferred Profit account of Islamic Bank of Jordan at the end of the 5th year after the payment of the 5th instalment but before redemption? a....
-
Instructions You are to complete a ten page, typed, double-spaced essay on one of the following: Gender Differences in Communication The paper should include relevant research on your topic. You...
-
Nowadays, company performance is not only measured by profit and loss in financial statement but also how well the company manages its: a. Environmental performance Diversity of its workforce Safety...
-
What are some of the malstressors that you have observed in a justice agency or organization? Describe the consequences of organizational stress in the agency or organization. What personality traits...
-
In the current year, Maria paid the following taxes: Special assessment to provide local benefits $2,500 County real estate taxes paid on her vacation home 1,250 Sales taxes paid when she purchased a...
-
Consider a damped oscillator with m = 0.2 kg, k = 100 N/m, and b = 5 N-s/m. The oscillator is driven by a force F= (1.6 N) cos 20r. (a) If the displacement is given by x = A cos(wt - 8), what are the...
-
Determine, based on Beamons description of the Ogive Funds characteristics, his likely inclination to aggressively implement the funds strategy. Justify your response. Morris next meets Robin Barker,...
-
The following exercises are not grouped by type. Solve each equation. x610x -9
-
Show that the criterion for chemical equilibrium developed in the text, for a closed system at constant temperature and pressure, is also the equilibrium condition to be satisfied for closed systems...
-
Determine whether these structures represent the same compound or isomers; a) H-C. -C -C H-C-H H H b) H-C-C. H-C- H H H H-C-H H H TI C_CH C H H H c) H=C=C d) H-C-C H H-CH H-C- ...
-
Calculate the DU for these formulas and draw two constitutional isomer for each: (a) C 10 H 22 (b) C 9 H 16 (c) C 6 H 6
-
Convert these structures to skeletal structures: T a) H-C-C H H H-C-H C c) CH3CHCCHCH(CH3)2 C=C=C-H b) H H-C-H H H CH3 d) CH,CHCH,NHCH,CH CH,
-
Answer the following questions with respect to the following Strategy pattern diagram as illustrated below 3. The relationship between StudentToSchool class and Strategy and actors with strategy?...
-
a) Design a simple relational database that captures the information required to maintain records of orders. Take into account that your design should allow a customer to order as many items as...
-
Write a procedure named AVG in assembly language which calculates and returns the average of three memory operands. Also show how this procedure will be called from main. Pass arguments (variable...

Study smarter with the SolutionInn App


