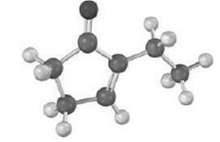
The following molecule was founded by an intermolecular aldol reaction. What dicarbonyl precursor was used for itspreparation?
Question:
The following molecule was founded by an intermolecular aldol reaction. What dicarbonyl precursor was used for itspreparation?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (15 reviews)
H 4Oxohe...View the full answer

Answered By

Jeff Omollo
As an educator I have had the opportunity to work with students of all ages and backgrounds. Throughout my career, I have developed a teaching style that encourages student engagement and promotes active learning. My education and tutoring skills has enabled me to empower students to become lifelong learners.
5.00+
5+ Reviews
42+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following molecule was formed by a Robinson annulations reaction. What reactants wereused?
-
What starting compound would you use in an aldol cyclization to prepare each of the following? (a) (b) (c)
-
The reversibility of the aldol addition reaction is a major factor in each of the following problems. Provide a plausible curved-arrow mechanism for each transformation. H2O OH
-
Dodona I, LLC, invested $ 4 million in two securities offerings from Goldman, Sachs & Co. The investments were in collat-eralized debt obligations ( CDOs). Their value depended on residential...
-
Consider a firm whose competitive advantage is built almost entirely on its ability to achieve economies of scale in producing small electric motors that are used by the firm to make hair dryers,...
-
Based on what you know about the following organizations, how would you categorize them according to Mintzbergs Five Organizational Types (Exhibit 1.7): General Electric? Facebook? Toyota Motor...
-
Explain the importance of maintaining complete and accurate records.
-
Compute the charitable contribution deduction (ignoring the percentage limitation) for each of the following C corporations. a. Amber Corporation donated inventory of clothing (basis of $24,000, fair...
-
Explain how you plan to gain awareness for your product(soft drink). Discuss your integrated marketing communications (IMC) plans including advertising, sales promotions, public relations, personal...
-
The Melitz effect. Open the spreadsheet ''heterogeneous firms.xis." This provides data for a hypothetical monopolistically competitive market with heterogeneous firms. Each firm is numbered from 1 to...
-
The following structure represents an intermediate formed by addition of ester emulation to a second ester molecule. Identify the reactant, the leaving group, and theproduct.
-
Which of the following compounds would you expect to undergo aldol self-condensation? Show the product of each successful reaction. (a) Trimethylacetaldehyde (b) Cyclopentanone (c) Benzophenone...
-
In Exercises 31 through 36, find all points (both coordinates) on the given curve where the tangent line is (a) Horizontal (b) Vertical. xy = 16y 2 + x
-
Kerstin Dugmore plans to send their kids to college in 4 years and will need another $13,420.00 before they can do so. Given an interest rate of 1.00% compounded tri-annually, how much money do they...
-
Liability insurance is First-party insurance. Fourth-party insurance. Third-party insurance. Second-party insurance. best answer?
-
Apply the Ke formula that you learned in the chapter to a stock from a US corporation that is currently trading. Use a constant growth rate of 4%. Explain what you have discovered
-
List and explain 2 methods of common forms of verifiable evidence that could be used to review customer satisfaction.
-
Asset allocation funds typically invest in Blank______. Multiple select question. money market instruments broadway plays bonds stocks real estate
-
If a taxpayer has $140,000 of employee salary, how much will be withheld for the Social Security and Medicare taxes in 2017?
-
Representative data read from a plot that appeared in the paper Effect of Cattle Treading on Erosion from Hill Pasture: Modeling Concepts and Analysis of Rainfall Simulator Data (Australian Journal...
-
Write Lewis structures for each molecule or ion. Use expanded octets as necessary. a. PF c. SF4 b. 13 d. Gel
-
Predict the products of E1 elimination of the following compounds. Label the major products. (a) (b) (c) , , Br (CHC-CH CH Br
-
Propose mechanisms and draw reaction-energy diagrams for the following reactions. Pay particular attention to the structures of any transition states and intermediates. Compare the reaction-energy...
-
Protonation converts the hydroxyl group of an alcohol to a good leaving group. Suggest a mechanism for each reaction. (a) (b) OH H,SO, heat (EI) + H,O OH Br HBr, heat 2 or S1 + H2O
-
Vanguard Corporation issued $8,000,000 of 10% bonds on September 1, 2023, due on September 1, 2029. The interest is to be paid twice a year on August 31 and February 28. The bonds were sold to yield...
-
If football matches were shortened form 90 minutes to 45 minutes, would you expect more uncertainty of outcome or less? Refer to any sports economic theories.
-
The Canadian Restaurant Association states that the restaurant industry has an economic effect of more than $1.7 trillion annually, with every dollar spent in restaurants generating an estimated...

Study smarter with the SolutionInn App


