The following reactions are unlikely to provide the indicated product in high yield. What is wrong witheach?
Question:
The following reactions are unlikely to provide the indicated product in high yield. What is wrong witheach?
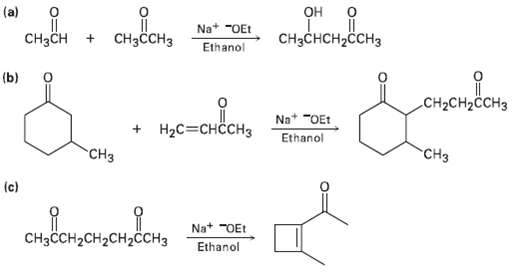
Transcribed Image Text:
о. одо, (a) сые еь он Na* "OEt CHснсH2CсHз CHзсCHз CHзCH Ethanol (b) .oвon CH2CH2CCH3 Na* "OEt + HаС3снсснз Ethanol "СHз "CHз (c) сононсн дсн Na+ "OEt CHзссH-CH2CH2CСH3 Ethanol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a Several other products are formed in addition to the one pictured ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
Which of the following reactions are possible, and by what interaction could they occur? For those forbiddevn explain why. (a) + p K0 + p + 0 (b) K + p 0 + 0 (c) K+ + n + + 0 + y (d) K + 0 + 0 + +...
-
Which of the following reactions are allowed, and which are forbidden, by the conservation laws appropriate to weak interactions? (a) + p + + n, (b) e + p e + + + p, (c) + e + e , (d) ...
-
Which of the following reactions are allowed and which are forbidden by the conservation laws appropriate to weak interactions? (a) + + e + e ; (b) K + + + e + e ; (c) K + 0 + + + ;...
-
Given the following network, with the indicated flow capacities along each branch, determine the maximum flow from source node 1 to destination node 10 and the flow along eachpath: 15 5 10 7 16 12 0...
-
Mercury is a hypothetical store that sells athletic shoes, particularly shoes for runners. Mercury is distinctive in the training of its sales staff. The store has a variety of diagnostic tools,...
-
If two electrons are each 1.50 x 10 -10 m from a proton, as shown in Fig. E 21.37, find the magnitude and direction of the net electric force they will exert on the proton. Figure E21.37 65.0
-
Describe the purpose of certification and licensure, and list reasons for revocation of licenses.
-
Write the structure of the major organic product formed in the reaction of 1-pentene with each of the following: (a) Hydrogen chloride (b) Hydrogen bromide (c) Hydrogen bromide in the presence of...
-
New Tech Corporation has assets with a market value of $300 million, $40 million of which are cash. It has debt of $50 million, and 7 million shares outstanding. Assume perfect capital markets and no...
-
Lazy River Resort opened for business on June 1 with eight air-conditioned units. Its trial balance before adjustment on August 31 is as follows. In addition to those accounts listed on the trial...
-
The so-called Wieland?Miescher ketone is a valuable starting material used in the synthesis of steroid hormones. How might you prepare it from 1, 3-cyclohexanedione? Wieland-Miescher ketone
-
Fill in the missing reagents a?h in the following scheme: CO2Et CH3 CO2Et 2. f 1. c 2. d 1. a 2. b 1. g 1. e 2. h CO2Et
-
In 2015, Addison Group (a provider of professional staffing services) and Kelton (a global insights firm) surveyed the work preferences and attitudes of 1,006 working adults spread over three...
-
Cash flows from investing activities are Select answer from the options below negative from selling long-term assets and negative from purchasing them. positive from selling long-term assets and...
-
If a firm's revenues and costs are denominated in the same currency, then they are Group of answer choices best off using forward contracts to lock in exchange rates naturally hedged best off using...
-
Define: Foreign currency. b. The exchange rate. The U.S. interest rate differential.
-
An employee who uses their own vehicle for employment purposes cannot deduct any financing costs related to the purchase of the car An employee who is provided with a vehicle owned by the employer...
-
Alanzo submits an application for a $300,000 whole life insurance policy with the help of his agent and also applies for coverage under a temporary insurance agreement (TIA). His health status is...
-
Thomas received $30,000 in a legal settlement in 2017. The tax treatment of the item is not certain. Thomass research results were ambiguous and he is not sure if the income is taxable. Because some...
-
Independent random samples of sizes n1 = 30 and n2 = 50 are taken from two normal populations having the means 1 = 78 and 2 = 75 and the variances 21 = 150 and 22 = 200. Use the results of Exercise...
-
Draw the Lewis structure for BrF with an arrow representing the dipole moment. Refer to Figure 10.10 to estimate the percent ionic character of the BrF bond. Percent ionic character 100 75 50 25 0....
-
Predict the major products of acid-catalyzed dehydration of the following alcohols. (a) Pentan-2-ol (b) 1-methylcyclopentanol (c) 2-methylcyclohexanol (d) 2,2-dimethylpropan-1-ol
-
Propose mechanisms for the following reactions. Additional products may be formed, but your mechanism only needs to explain the products shown. (a) (b) (c) OEt EtOH heat Br OEt CH2OH H SO4 heat...
-
Predict the dehydrohalogenation product(s) that result when the following alkyl halides are heated in alcoholic KOH. When more than one product is formed, predict the major and minor products. (a)...
-
Image transcription text You encounter a split system that uses R-22 refrigerant and observe the following refrigeration parameters from the unit's control display. The unit is operating in cooling...
-
Calculating Present Values Imprudential, Inc., has an unfunded pension liability of $645 million that must be paid in 25 years. To assess the value of the firm's stock, financial analysts want to...
-
Describe the operating activities of each company noting similarities and difference between COCA COLA & PEPSICO. Identify two economy wide factors and industry wide factors that could impact on the...

Study smarter with the SolutionInn App


