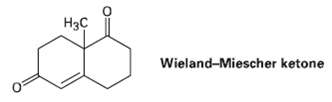
The so-called Wieland?Miescher ketone is a valuable starting material used in the synthesis of steroid hormones. How
Question:
The so-called Wieland?Miescher ketone is a valuable starting material used in the synthesis of steroid hormones. How might you prepare it from 1, 3-cyclohexanedione?
Transcribed Image Text:
Нас Wieland-Miescher ketone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
S 1 NatOEt H3C EtOH S 2 CHI 1 Na OEt EtOH 2 HCCHCO...View the full answer

Answered By

Ajeet Singh
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life.
I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge.
I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields.
Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a teacher. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
4.90+
7+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How might you prepare the following compounds using an alkylation reaction as the keystep? (b) -H CH-CH (a) CH2CH=CH2 (c) CHCH,CH2CHCEN C (d) (f) (e) CH - CH CHCHCHCH CH-CH - Hg, CH(CH3)2
-
How might you prepare Pentylamine from the following starting materials? (a) Pentanamide (b) Pentanenitrile (c) 1-Butene (d) Hexanamide (e) 1 -Butanol (f) 5-Decene (g) Pentanoic acid
-
How might you prepare 2-phenylethanol from benzyl bromide? More than one steps inneeded. CH2Br CH2CH2OH
-
Julia Robertson is a senior at Tech, and she's investigating different ways to finance her final year at school. She is considering leasing a food booth outside the Tech stadium at home football...
-
Which of the following circumstances are likely to create early-mover advantages? a) Maxwell House introduces the first freeze-dried coffee. b) A consortium of U.S. firms introduces the first...
-
The pV-diagram in Fig. 20.5 shows a cycle of a heat engine that uses 0.250 mole of an ideal gas having y = 1.40. The curved part a of the cycle is adiabatic. (a) Find the pressure of the gas at point...
-
Describe the importance of a multidisciplinary approach to patient care.
-
1. Are the auditors guilty of malpractice? Explain. 2. What defenses are available to Joker & Wild in this case? Explain what they must prove to successfully assert these defenses. 3. Assume you are...
-
The Rando Corporation's stock has a beta of 1.5. If the excess return on the stock market increases by 5%, by approximately how much will the excess return on Rando Corporation's stock increase?
-
Joey Cuono started his own consulting firm, Cuono Company SpA on June 1, 2017. The trial balance at June 30 is shown below. In addition to those accounts listed on the trial balance, the chart of...
-
How might the following compounds be prepared using Michael reactions? Show (he nucleophilic donor and the electrophilic acceptor in eachcase. ", , (b) . (a) CHCCH-CH2CH2CH3 CH2H2CCgHs o2Et NO2 (d)...
-
The following reactions are unlikely to provide the indicated product in high yield. What is wrong witheach? . , (a) Na* "OEt CHH2CH CHCH CHCH Ethanol (b) .oon CH2CH2CCH3 Na* "OEt + H3 Ethanol "H...
-
Complete this table for H2O: T, F P psia u, Btu/lbm Phase description 300 782 40 120 400 Saturated liquid 500 400
-
Starbucks' decision to buy out its joint venture partner in Japan following several years of operation and instead establish a wholly owned subsidiary Multiple Choice suggests that the company no...
-
The fixed-charge coverage ratio requirements limits a company's ability to Multiple choice question. repurchase stock. enter into a merger with another company. pay off other debts. pay dividends.
-
Loan charge-offs for banks Multiple choice question. decrease net income. have no impact on invested capital. increase invested capital. decrease invested capital.
-
The risk in bond funds is related to changes in: Multiple choice question. real estate prices interest rates stock market prices
-
Accounting: financial statment analysis individual company information For your corporation Anheiser Bush, write two paragraphs describing the business operations, include a brief history, risks and...
-
Alison accidentally omitted $40,000 of gross income from the restaurant she owned on her 2016 tax return. The return showed gross income of $150,000 when filed on October 15, 2017. When can the IRS...
-
CLASS PERIO Solving Linear Equations: Variable on Both Sides Solve each equation. 1) 6r+ 7 = 13 + 7r 3) -7x-3x+2=-8x-8 5)-14 +66+7-26=1+5b 7) n-3n = 14-4n 2) 13-4x=1-x 4)-8-x= x - 4x 6)n+2=-14-n 8)...
-
Write the Lewis structure for each molecule or ion. a. H3COH b. OH- c. Bro- d. 0-
-
Predict the products formed by sodium hydroxide-promoted dehydrohalogenation of the following compounds. In each case, predict which will be the major product. (a) 1-bromobutane (b) 2-chlorobutane...
-
What halides would undergo dehydrohalogenation to give the following pure alkenes? (a) Hex-1-ene (b) Isobutylene (c) Pent-2-ene (d) Methylenecyclohexane (e) 4-methylcyclohexene
-
In the dehydrohalogenation of alkyl halides, a strong base such as tert-butoxide usually gives the best results via the E2 mechanism. (a) Explain why a strong base such as tert-butoxide cannot...
-
Image transcription text Wind tunnel measurements of the pressure and skin friction around a NACA 2415 airfoil at 8 degrees angle of attack resulted in the following data of pressure and skin...
-
Image transcription text The following table contains load-extension data from a tensile test on a cylindrical specimen with gauge length 9mm and gauge diameter 5mm. Load-extension Data Load [KN] 0...
-
Image transcription text Systems Modelling and Analysis - Assignment 1 Due: Friday 25/08/2022 by 5:00:00 pm. To be submitted individually on Canvas and Gradescope. Part 1: Dartboard Positioning...

Study smarter with the SolutionInn App


