The following thermal isomerization occurs tinder relatively mild conditions. Identify the pericyclic reactions involved, and show how
Question:
The following thermal isomerization occurs tinder relatively mild conditions. Identify the pericyclic reactions involved, and show how the rearrangementoccurs.
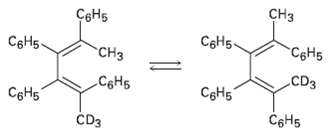
Transcribed Image Text:
CHз СвНь CоНб- CeH5. C&H5 "CHз CDз CoH5 CgH5 CEH5 СоНs CDз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
The diene can cyclize by either of two conrotatory paths to form cyclobutenes A and B D3C D...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following isomerization reaction occurs in the liquid phase: A B, where A and B are miscible liquids for which: GE/RT = 0.1 xAxB- If Go298 = -1,000 J, what is the equilibrium composition of the...
-
Show how Diels-Alder reactions might be used to synthesize the following compounds. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH 3COOCH CH3 CN CN CI CI Cl Cl Cl CI CI C chlordane CI CI Cl Cl CI C aldrin CN...
-
Show how you would use Suzuki reactions to synthesize these products from the indicated starting materials. You may use any additional reagents you need. (a) (b) Br
-
5.2-1 Dielectric Media. Identify the media described by the following equations, regarding linearity, dispersiveness, spatial dispersiveness, and homogeneity. (a) = x - av x 8, (b) + a2 = (c) a, 8/a1...
-
Is the highest bid necessarily the best bid? Explain your answer.
-
Use the data in CARD.RAW for this exercise. (i) The equation we estimated in Example 15.4 can be written as log(wage) = 0 + 1 educ + 2 exper + ... + u, where the other explanatory variables are...
-
The van der Waals equation of state is applicable for the (a) Solid phase only (b) Liquid phase only (c) Liquid and gas phases only (d) Solid, liquid and gas phases.
-
1. How does the post-closing holding company structure protect the interests of the financial sponsor group, creditors, and the utilitys customers, employees, and other stakeholder groups? 2. What...
-
At April 30, Blossom Company has the following bank information: Cash balance per bank $8100 Outstanding checks $500 Deposits in transit $975 Credit memo for interest $20 Bank service charge $40....
-
Holmes Corporation is a leading designer and manufacturer of material handling and processing equipment for heavy industry in the United States and abroad. Its sales have more than doubled, and its...
-
What stereochemistryantarafacial or suprafacialwould you expect to observe in the following reactions? (a) A photo chemical [1, 5] sigma tropic rearrangement (b) A thermal [4 + 6] cyclo addition (c)...
-
Would you expect the following reaction to proceed in a conrotatory or disrotatory manner? Show the stereochemistry of the Cyclobutene product, and explain youranswer. hv
-
Describe three uses for a read-only database.
-
Discuss the elements a contract must have in order to be legally binding, referring to each element describe an experience engaging in a contract with another party, was the contact legally binding...
-
TR's ageing department actually consists of two processes. First, for some batches, various dyeing agents are applied to the jeans. Next, the jeans are run through a tumbler to soften, and age the...
-
Waivers protect service providers from liability for injuries that occur because of what type(s) of risks?
-
Assume Vlad is presented with an opportunity from another tech company to develop software for a music sharing platform. By coincidence, a portion of the code he developed for the social media app...
-
+ Watch the video and then provide answers on the righthand side of the column for easy grading. Does Kentucky have a Safe Haven Yes, there are 16 Kentucky locations. Baby Box? What individual(s) can...
-
Some flowering plants rely on insects to carry pollen from male flowers to female flowers. The insects are dusted with pollen as they drink nectar produced by the flowers. Is this an example of...
-
Suppose that you could invest in the following projects but have only $30,000 to invest. How would you make your decision and which projects would you invest in? Project Cost $ 8,000 11,000 9,000...
-
Some stock brokerage firms have offered temporary specials where they dont earn commissions on stock trades and a few (e.g., Robinhood Financial, discussed in the chapter) have made zero stock...
-
A single organic product was obtained when 1-bromo-3-chloropropane was allowed to react with one molar equivalent of sodium cyanide in aqueous ethanol. What was this product?
-
Each of the reactions shown involves nucleophilic substitution. The product of reaction (a) is an isomer of the product of reaction (b). What kind of isomer? By what mechanism does nucleophilic...
-
Arrange the isomers of molecular formula C4H9Cl in order of decreasing rate of reaction with sodium iodide in acetone.
-
An airline will purchase 2 million gallons of jet fuel in one month and hedges using heating oil futures. From historical data, the standard deviation of heating oil futures contract is sF =0.031...
-
A European call option for a share costs $5.00. The exercise price of the call option is $100.00. An investor buys one call and holds it until maturity: a. Under what circumstances will the holder of...
-
You have been asked to estimate the cost of capital for the UTX corporation. The company has 7 million shares and 150,000 bonds outstanding at par value $10,000. In addition, it has $300 million in...

Study smarter with the SolutionInn App


