The hydration of an alkyne is not a reasonable preparative method for each of the following compounds.
Question:
The hydration of an alkyne is not a reasonable preparative method for each of the following compounds. Explain why.
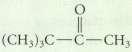
Transcribed Image Text:
(CH)C C CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 92% (14 reviews)
Hydration can only be used to prepare ketones tha...View the full answer

Answered By

Shem Ongek
I am a professional who has the highest levels of self-motivation. Additionally, I am always angled at ensuring that my clients get the best of the quality work possible within the deadline. Additionally, I write high quality business papers, generate quality feedback with more focus being on the accounting analysis. I additionally have helped various students here in the past with their research papers which made them move from the C grade to an A-grade. You can trust me 100% with your work and for sure I will handle your papers as if it were my assignment. That is the kind of professionalism that I swore to operate within. I think when rating the quality of my work, 98% of the students I work for always come back with more work which therefore makes me to be just the right person to handle your paper.
4.80+
174+ Reviews
426+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Write an equation for the synthesis of 2-octanone by a. Oxidation of an alcohol b. Hydration of an alkyne
-
The standard free energy of activation (G++) for hydration of 2-methylpropene to 2-methyl-2-propanol (Eq. 4.41, p. 169) is 91.3 kJ mol-t (Zt 8 kcal mol-l;. The standard free energy G for hydration of...
-
Hydration of alkynes (via oxymercuration) gives good yields of single compounds only with symmetrical or terminal alkynes. Show what the products would be from hydration of each compound. (a)...
-
Establish procedures to guarantee substantiation of claims for allowances. Think about how you would set up a process to ensure employee claims and allowances could be claimed. Write a step by step...
-
MMC maintains a qualified defined benefit plan for eligible employees, with an effective date of January 1, 1990. The plan year for vesting and participation purposes is the calendar year....
-
The figure shows a finger exerciser used by law-enforcement officers and athletes to strengthen their grip. It is formed by winding A227 hard-drawn steel wire around a mandrel to obtain 2 turns when...
-
A partial condenser operates as shown in Figure 10.36. Assuming that \(T_{0}=70^{\circ} \mathrm{F}\), calculate the following: (a) Condenser duty (b) Change in availability function (c) Lost work (d)...
-
Founded in 1983, the Beverly Hills Fan Company is located in Woodland Hills, California. With 23 employees and sales of less than $10 million, the company is relatively small. Management feels that...
-
What are some of the factors that make it difficult for unions to coordinate their efforts across national borders? What is a great personal branding statement for a cyber-security human resources...
-
Since London is north of Paris and south of Edinburgh, it follows that Paris is south of Edinburgh. The following arguments are deductive. Determine whether each is valid or invalid, and note the...
-
Draw a Lewis structure for each of the following alkynes. (a) isopropylacetylene (b) cyclononyne (c) 4-methyl-1-pentyne
-
Compare the results of hydroboration-oxidation and mercuric ion-catalyzed hydratioa for 2-butyne.
-
Establishing a culture of sound business ethics within an organization is challenging, to say the least. Companies that market products which are not considered to be "healthy" for consumers have...
-
The following picture shows UCLA and Downtown Los Angeles as observed from the Getty Center. The smog in this picture is so that we can barely see the bottom of the high rise buildings in downtown....
-
A point-like particle is moving along a straight line. Alice and Bob are two observers and they respectively measure the motion of the particle in two reference frames called "xAy" and "XBY" with the...
-
A periodic function f(t) is defined as i) f(t)=t +, -, f(t) = f(t+2) -
-
Solve for the minimum cost using transportation method and computation for improvement. I X Y Z Demand 1 20 10 5 4 3 2 15 3 3 3 3 10 3 3 4 Supply 12 13 20
-
The mass of Link 1 and 2 of the manipulator as in Problem 1 is 1.0 kg and 0.6 kg, respectively, all mass exists as a point mass at the distal end of each link, as describe in Section 6.7, textbook...
-
Refer to the data on malt extract in Table D. 8 of the Data Bank. (a) Obtain sample mean and standard deviation. (b) Obtain quartiles. (c) Check conformity with the empirical rule. Data from in Table...
-
Test whether the 5-year survival rate for breast cancer is significantly different between African American and Caucasian women who are younger than 50 years of age and have localized disease....
-
Answer the following questions for the 2,4,6-heptatrienyl cation. (a) Which MO is nonbonding? (b) Classify each MO as symmetric or antisymmetric. (c) To which carbon atoms in this cation is the...
-
Classify the following sigmatropic reaction by giving its bracketed-number designation and its stereochemistry with respect to the plane of the -electron system. CH3 CH3 H S configuration
-
(a) Explain why two monomethyl esters of N-acetyl-laspartic acid are known. Draw their structures. (b) Explain why a mixture of these two compounds can be separated by cation-exchange chromatography...
-
Career pathing programs arm staff with skills and provides incentives of growth, challenge, and visibility. It presents the chance to change the way a technology team is perceived internally and...
-
In order to better understand what you are trying to communicate, does Christianity and the Way of Jesus fall under a honor society, justice society or both?
-
For HOST280, I have a significant assignment. Core Values I need to be able to define what each core value means. To define these three core values. The definition is personal so that if someone asks...

Study smarter with the SolutionInn App


