Classify the following sigmatropic reaction by giving its bracketed-number designation and its stereochemistry with respect to the
Question:
Classify the following sigmatropic reaction by giving its bracketed-number designation and its stereochemistry with respect to the plane of the π-electron system.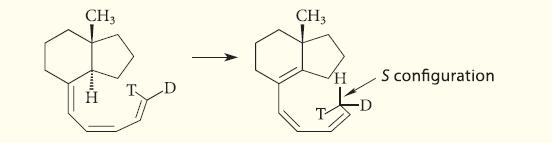
Transcribed Image Text:
CH3 CH3 H S configuration
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
First identify the bond that is migrating Because the hydrogen atom migrates one end of the migratin...View the full answer

Answered By

Vijesh J
My passion to become a tutor is a lifetime milestone. Being a finance and marketing professional with hands-on experience in wealth management, portfolio management, team handling and actively contributing in promoting the company. Highly talented in managing and educating students in most attractive ways were students get involved. I will always give perfection to my works. Time is the most important for the works and I provide every answer on time without a delay. I will proofread each and every work and will deliver a with more perfection.
4.70+
5+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Refer to Study Problem 28.2 and, assuming an antarafacial migration, give the structure of a starting material that would give a stereoisomer of the product with the R configuration at the...
-
Googles ease of use and superior search results have propelled the search engine to its num- ber one status, ousting the early dominance of competitors such as WebCrawler and Infos- eek. Even later...
-
Classify the following sigmaffopic reactions with bracketed number.
-
Kim and Kanye have been dating for years and are now thinking about getting married. As a financially sophisticated couple, they want to think through the tax implications of their potential union....
-
Elizabeth Pie Company has been in business for 50 years and has developed a large group of loyal restaurant customers. Giant Bakery Inc. has made an offer to buy Elizabeth Pie Company for $5,000,000....
-
Nelson Enterprises exchanged a building it owned in Grand Junction for a building in Canon City owned by Lamb Corporation. The buildings were both valued at $ 575,000, so there was no cash...
-
One critical-thinking skill is a heightened awareness of the danger of reaching a conclusion prior to acquiring missing information that were it known would have a reasonable probability of altering...
-
Central Station Fund. The Town of Elizabeth operates the old train station as an enterprise fund. The train station is on the national register of historic buildings. Since the town has held the...
-
If your car gets 25 miles per gallon, how much does it cost to drive 420 miles when gasoline costs $2.50 per gallon? The cost is $ (Simplify your answer. Round to the nearest cent as needed.)
-
Answer the following questions for the 2,4,6-heptatrienyl cation. (a) Which MO is nonbonding? (b) Classify each MO as symmetric or antisymmetric. (c) To which carbon atoms in this cation is the...
-
(a) Explain why two monomethyl esters of N-acetyl-laspartic acid are known. Draw their structures. (b) Explain why a mixture of these two compounds can be separated by cation-exchange chromatography...
-
Cocoa Beach Hotels and Resorts had the following transactions during the current period: Feb. 22 Issued 10,000 shares of $1 par value common stock to attorneys in payment of a bill for $30,000 for...
-
Q. No. 5. In the current year, Phillip Hard earned the following income: Employment income Property income Gains: Shares of Corporation X $12,000 Personal-use property $80,000 2,000 7,000 Listed...
-
Providers of zero-rated goods are not allowed to claim input tax credits ? explain why
-
You have decided that instead of buying a car when you go to college that you will join Flexcar. To join Flexcar, you must pay an annual fee of $35. Then, when you need to use a car you pay $55 a day...
-
Jhudora owns one rental property. She actively participates in managing the property, including identifying tenants and arranging for maintenance. She has suspended losses of $13,000 with respect to...
-
What strategies would you use for dealing with the following Client Concerns: Anxiety Confusion Communication Understanding Stress Uncertainty
-
The following are three decisions that Joel Stager needs to make. Joel manages an up-market restaurant in Miami, Florida, and has considerable discretion over its operations. Decision 1: Whether to...
-
Write a paper about the Working relationship in the organization- collaboration within and outside the organization
-
Rank the following compounds in order of increasing basicity. CH OH NH
-
In the following set, arrange the compounds in order of decreasing pKa, and explain your reasoning. 0 OCH, OCH3
-
Rationalize your answer to the previous problem by explaining why more energy is required to ionize acetic acid than fluoroacetic acid. Previous Problem 3.22 Calculate the standard free energy for...
-
(2) Identify the mission statement and purpose for the agency you have selected. The mission statement may not be presented in the document you downloaded, be sure to check the agency website to...
-
Landing Page for Online Media Segmentation You are building an Online African Media Segmentation that includes Online TV Stations, Online Radio Stations, and online press. You are asked to provide...
-
Why do Companies and businesses use job performance evaluation to find how well their employees perform in their respective duties?. Thorough explanation with references. Thank you

Study smarter with the SolutionInn App


