The Knoevenagel reaction is a carbonyl condensation reaction of an ester with an aldehyde or ketone to
Question:
The Knoevenagel reaction is a carbonyl condensation reaction of an ester with an aldehyde or ketone to yield an ?, ??un-saturated product. Show the mechanism of the Knoevenagel reaction of diethyl malonate with benzaldehyde.
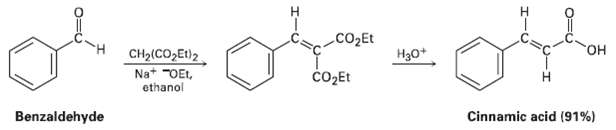
Transcribed Image Text:
cO2Et H30+ HO. CH2(CO,Et)2 čO2Et Na+ "OEt, ethanol Cinnamic acid (91%) Benzaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Formation of the enolate of diethyl malonate is the ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
An ester is a compound formed by a condensation reaction between a carboxylic acid and an alcohol. Read the discussion of esters in Section 24.4 and then give an example of a reaction forming an...
-
The reaction of an aldehyde or ketone with a Grignard reagent is a nucleophilic addition to the carbon-oxygen double bond. (a) What is the nucleophile? (b) The magnesium portion of the Grignard...
-
Diethyl malonate is prepared commercially by hydrolysis and esterification of ethyl cyanoacetate. The preparation of ethyl cyanoacetate proceeds via ethyl chloroacetate and begins with acetic acid....
-
We look at the accumulated area beneath this curve, as in the definite integral as follows F(x) = f(t) dt -2 { F(x) = int_(-2)^x f(t) dt a) Use ordinary area formulas to compute each of the...
-
Give examples of actions that are substitutes in production. Give examples of actions that are compliments in production.
-
The following data represent the chemistry grades for a random sample of 12 freshmen at a certain college along with their scores on an intelligence test administered while they were still seniors in...
-
Understand and describe common medical errors and how they lead to litigation.
-
Refer to the information presented in Mini-Exercise 6.3. Assume that Gandolfi Construction Co. calculated depreciation expense for the CAT 336DL earth mover on the straight-line method and reported...
-
What would be the gross debt service (GDS) ratio based on the following information? Current monthly gross income = $4000 Current monthly after-tax income = $2800 Monthly mortgage payment = $950...
-
A rocket motor is manufactured by bonding together two types of propellants, an igniter and a sustainer. The shear strength of the bond y is thought to be a linear function of the age of the...
-
The amino acid leucine is biosynthesized from a-ketoisovalerate by the following sequence of steps. Show the mechanism ofeach. Acetyl CoA COASH NAD+ NADH/H* CO2 "C 2-Isopropylmalate co2 ...
-
The Darzens reaction involves a two-step, base-catalyzed condensation of ethyl chloroacetate with a ketone to yield an epoxy ester. The first step is a carbonyl condensation reaction, and the second...
-
A vector has the components Ax = 22 m and Ay = 13 m. (a) What is the magnitude of this vector? (b) What angle does this vector make with the positive x axis?
-
the report gives credence of to the auditor and general of South Africa (AGSA) findings that the eight metropolitans municipalities ,44 district municipalities and estimated 200 local municipalities...
-
Transaction Description: Waren Sports Supply received a partial payment of $12,000 from eastern Wisconsin University for the balance owed after reducing the amount by the sales returned goods on...
-
A problem that has hindered the ESG movement to-date is: Question 3 Answer a. After the failure of Enron, no one trusts accountants b. Shareholders only care about maximizing profit c. No one knows...
-
Year 9 Income Statement Data in 000s Total Revenues $610,000 Cost of Goods Sold 370,000 Delivery Costs 40,000 Marketing Costs 48,000 Administrative Expenses 10,000 Operating Profit (Loss) 142,000...
-
Lala is a CPA who just received her degree and has a focus on corporate tax. She wants to work for a company (not an accounting firm) that will allow her to focus on corporate tax and get extensive...
-
Jennifer, age 35, is single and an active participant in her employers qualified retirement plan. Compute the maximum Roth IRA contribution that she can make in 2017 if a. her adjusted gross income...
-
The Taylor's series expansion for cosx about x = 0 is given by: where x is in radians. Write a user-defined function that determines cosx using Taylor's series expansion. For function name and...
-
Use the BornHaber cycle and data from Appendix IIB, Chapter 9, and this chapter to calculate the lattice energy of KCl. (H sub for potassium is 89.0 kJ/mol.) Substance Aluminum Al(s) Al(g) Al+(aq)...
-
Give the expected major product for each reaction, including stereochemistry where applicable. (a) but-1-ene + H2 / Pt (b) cis-but-2-ene + H2 / Ni (c) (d) + H/Pt + excess H,/Pt
-
One of the principal components of lemon grass oil is limoneneC10H16 When limonene is treated with excess hydrogen and a platinum catalyst, the product is an alkane of formula C10H20. What can you...
-
The chiral BINAP ligand shown in Figure 8-7 contains no asymmetric carbon atoms. Explain how this ligand is chiral.
-
Assume an organization needs to do mass layoffs in order to counteract the slowing market demand for their product. What would be considered going above and beyond legal duty to do the right thing by...
-
We are in an incredibly stressful time in our lives. Watch the Ted Talk, "How Burnout Makes Us Less Creative". Share your thoughts on the video. Then discuss what are some actions that you can take...
-
Image transcription text QUESTION 1 Diffusion (a) The diffusion coefficients for carbon in nickel are given at two temperatures as shown in Table 1: Table 1 diffusion coefficients for carbon in...

Study smarter with the SolutionInn App


