The mass spectra of 3-ethyl-2-pentanone and 4-methyl-2-pentanone are as follows. Explain which spectrum goes with which compound,
Question:
The mass spectra of 3-ethyl-2-pentanone and 4-methyl-2-pentanone are as follows. Explain which spectrum goes with which compound, what is the structure of the ion responsible for the peak at m/z 43 in eachspectrum?
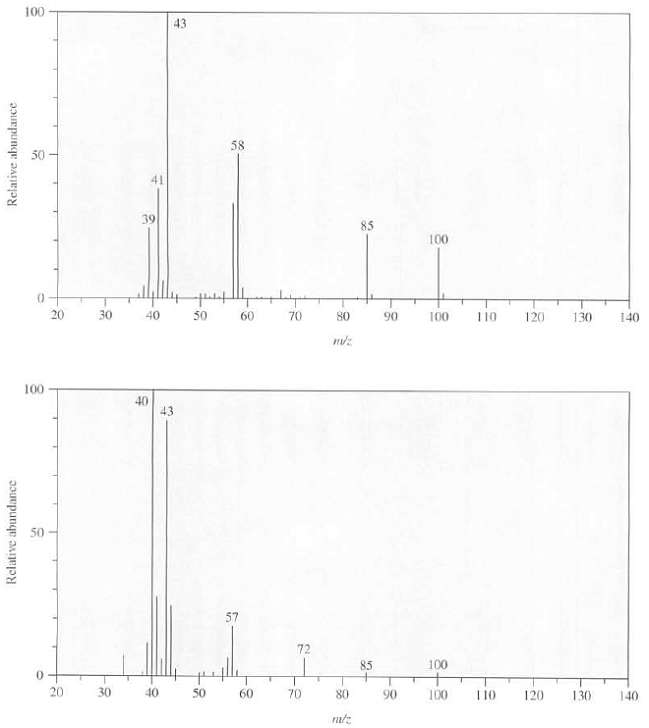
Transcribed Image Text:
100 43 58 50 41 39 85 100 20 30 40 50 70 60 80 90 100 110 120 130 140 100 40 43 57 72 100 85 20 30 40 50 60 70 80 90 100 T10 120 130 140 Relative abundance Relative abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
Both the ketones produce fragment ions at mz 43 and 85 due t...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The mass spectra of 1-methoxybutane, 2-methoxybutane, and 2-methoxy-2-methylpropane are shown in Figure 13.7. Match the compounds with the spectra. 100 73 80 S 60 57 20 0 10 20 30 40 50 60 70 80 90...
-
The mass spectra of two very stable cycloalkanes both show a molecular ion peak at m/z = 98. One spectrum shows a base peak at m/z = 69, the other shows a base peak at m/z = 83. Identify the...
-
The mass spectra of acid derivatives follow the principles shown in Chapter 18 for other carbonyl compounds and for alkoxy groups. Both McLafferty rearrangements and alpha-cleavages are common. The...
-
Holly needs $21,800 worth of new equipment for his shop. He can borrow this money at a discount rate of 11% for a year. Find the amount of the loan Holly should ask for so that the proceeds are...
-
IBM has a bond issue outstanding with 14 years to maturity. When originally issued the bond had a par value of $1,000, a stated coupon rate of 12% and 15 years to maturity. Currently, similar risk...
-
A diesel engine has air before compression at 280 K, 85 kPa. The highest temperature is 2200 K and the highest pressure is 6 MPa. Find the volumetric compression ratio and the mean effective pressure...
-
What is a SOI?
-
On December 31, 2016, the ledger of Hernandez Company contained the following account balances: All the accounts have normal balances. Journalize the closing entries. Use 4 as the general journal...
-
Kitty is paid $1200 weekly, also receives the following in addition to her regular salary each pay: a group term life insurance non-cash taxable benefit of $15 and a taxable cash car allowance of...
-
The objectives of the experiment: Materials required: 1- The preparation of Al(acac), 2- The preparation of K;[Al(OX);] Name the products: 1- Al(acac); 2-K[Al(OX);] . Procedure: 2 4
-
Suggest a structure for the compound whose mass spectrum is asfollows: 100 107 81 93 188 70 80 110 120 90 100 130 150 160 170 140 180 190 200 Relative abundance
-
Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm ?1 in its IR spectrum and B has a peak at 1715cm ?1 , the mass spectra of A and B are as follows, show the structures of...
-
Solve each of these equations for 0 x 360. a. 2 sinx = cos x b. 2 sinx 3cos x = 0 c. 4sinx + 7 cos x = 0 d. 3cos2x 4sin2x = 0
-
Using which one (1) of the following investing strategies would be INCONSISTENT with a belief in the "Weak Form" of the "Efficient Market Hypothesis"? a. A Bottom-Up Approach b. Technical Analysis c....
-
For the second half of the semester, you will be working in the role of student intern at the Conestoga Consulting Company, does a lot of consulting for Conestoga College and companies near the...
-
Could you elaborate on the concept of germline mutations and their significance in hereditary diseases and evolutionary biology ?
-
Discuss in a short paragraph (3-5+ sentences) how the theory of the diffusion of innovation does or does not apply to your communication campaign.
-
Biological denitrification of nitrate-containing waste waters can be described by the follow- ing overall reaction. NO+a CH,OH+H bC,H,NO+cN+d CO+e HO a. Determine a, b, c, d, and e, if Yys= 0.5 g X/g...
-
Simulate the process in Eqs. (13.3) and (13.4) in the vicinity of the point at (X,0) and try to use as an early warning signal the logarithmic time dependence considered in Eq. (1) of [243]. Equation...
-
When an electric field is applied to a shallow bath of vegetable oil, why do tiny bits of thread floating in the oil align with the field like compasses in a magnetic field?
-
In late November and early December 2006, there was speculation in the financial markets as to what the Federal Reserve planned to do with interest rates. After raising the federal funds interest...
-
Trichloromethane (CHCl3, also called chloroform) has a larger dipole moment than CFCl3. Use three-dimensional structures and bond moments to explain this fact.
-
Indicate the direction of the important bond moments in each of the following compounds (neglect C-H bonds). You should also give the direction of the net dipole moment for the molecule. If there is...
-
Write structural formulas for all of the alkenes with (a) The formula C2H2Br2 and (b) The formula C2Br2Cl2. In each instance designate compounds that are cis-trans isomers of each other. Predict the...
-
Suppose the normalized pulse shape, h(t), shown below, is used to generate the pulse train v(t) = vnhr(t - nT) N=- where {n} are the transmitted symbols, with each Un selected independently from {-1,...
-
The following information is available for Bonita's Chocolates: Actual production 2,750 boxes Budgeted production 3,450 boxes Direct Materials Standard Actual 4 pounds of chocolate per box @ $6.00...
-
James is currently on a bridge, 2 1 meters above the water and throws a ball in the horizontal direction. The ball gets to the water surface at a horizontal distance of 1 2 meters from James. ( a )...

Study smarter with the SolutionInn App


