The O ? H hydrogen in acetic acid is much more acidic than any of the C
Question:
The O ? H hydrogen in acetic acid is much more acidic than any of the C ? H hydrogens. Explain this result using resonance structures.
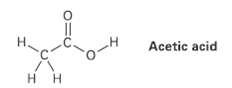
Transcribed Image Text:
Acetic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
H 0 HC 0 HH 0 HH The OH hydrogen of acetic acid i...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The pKa of the starting material (Ar-OH) is ~9, which is much more acidic than a normal alkyl alcohol (pKa ~15)? The reason is resonance stabilization of the conjugate base. Please draw the...
-
Pyrrole is much more acidic than diisopropylamine. Explain. pka = 16 pKa %3D pka = 36 Z-I Z-
-
One of the following hydrocarbons is much more acidic than the others. Indicate which one, and explain why it is unusually acidic.
-
A business has the following transactions: The business is started by receiving cash from an investor in exchange for common stock $20,000 The business purchases supplies on account $500 The...
-
Sally owns real property for which the annual property taxes are $9,000. She sells the property to Shelley on March 1 for $550,000. Shelley pays the real property taxes for the entire year on October...
-
If 500 newborns are screened at the inner-city hospital, then what is the exact binomial probability of exactly 5 HIV-positive test results? Infectious Disease Newborns were screened for human...
-
For each of the following situations, calculate the \(t\)-statistic \((t)\) : a. \(\mathrm{X}^{-}=20.00 ; \mu=18 ; s \mathrm{X}^{-}=1.00\) b. \(X^{-}=20.00 ; \mu=13 ; s X^{-}=1.00\) c. \(X^{-}=12.00...
-
An electronically excited state of Hg can be quenched by N, according to Hg* (g) + Nz (g, V = 0) -7 Hg (g) + N, (g, v = 1) in which energy transfer from Hg* excites Nz vibrationally. Fluorescence...
-
Locate the AICPA Code of Professional Conduct under module and go specifically to section 1.200 Independence. Please identify what you chose and share any real world examples you can find regarding...
-
Selecting the Best Two to Four Warehouse Locations for Als AthleticsMini Case Study. This hands-on example will give you your first opportunity to further your understanding of the books concepts by...
-
Alcohols can act either as weak acid or as weak bases, just as water can. Show the reaction of methanol, CH 3 OH, with a strong acid as HC1 and with a strong base such as NA + - NH 2 .
-
Write the products of the following acid-base reactions: (a) CH3OH + H2SO4 ? (b) CH3OH + NaNH2 ? (c) CH3NH3 + C1- +NAOH ?
-
Solve each equation in Exercises 114 by factoring. 2x(x 3) = 5x - 7x
-
How do long non-coding RNAs (lncRNAs) modulate gene expression through diverse mechanisms such as chromatin remodeling, transcriptional interference, and mRNA stability regulation, and what are the...
-
def hailstone(num): """ >>> hailstone(10) [10, 5, 16, 8, 4, 2, 1] >>> hailstone(1) [1] >>> hailstone(27) [27, 82, 41, 124, 62, 31, 94, 47, 142, 71, 214, 107, 322, 161, 484, 242, 121, 364, 182, 91,...
-
For each scenario below calculate the impact on cash, availability and net liquidity (cash + availability) Eligible AR of $100 is collected Raw material of $80 is received, eligible AR of $50 is...
-
In this assignment I will be using the dataset released by The Department of Transportation. This dataset lists flights that occurred in 2015, along with other information such as delays, flight time...
-
3. Create a folder in HDFS under the root directory called BigDataFirstName where "FirstName" is your name. 4. Copy the text file you created in step 2 from the Linux local file system into the HDFS...
-
Is it more important for theft offenses to be classified as specific-intent crimes than robbery? Why?
-
Derive Eq. (18.33) from Eq. (18.32).
-
Steam is produced at 70 bar and some unknown temperature. A small amount of steam is bled off just before entering a turbine and goes through an adiabatic throttling valve to atmospheric pressure....
-
Indicate whether each of these objects is chiral or achiral: (a) Golf ball (b) Baseball glove (c) Clock (d) T-shirt (e) Dress shirt (f) Automobile
-
Determine whether each of these molecules is chiral, for those that are chiral, put an asterisk at the chirality center. b) a) d) I e) )
-
Indicate whether each of these objects or molecules has a plane of symmetry: c) Ear b) Pencil a) Idealized human face I CH3 e) f) d) CH3 . Cl Br CH3 "H. CH3 "H g) h) Cl
-
Pharoah Company has actual sales of $54,700, flexible budget sales of $54,330, and master budget sales of $52360. Compute the sales price variance and sales activity variance (simplified) for Pharoah...
-
The first-quarter tax return needs to be filed for Prevosti Farms and Sugarhouse by April 15, 2022. For the taxes, assume the second February payroll amounts were duplicated for the March 4 and March...
-
Nico issues $10 million, 6%, 5 year bonds on January 1 of FY 1. The bonds pay interest semiannually on June 30 and December 31. The effective interest or market rate is 5%. What are the bond proceeds...

Study smarter with the SolutionInn App


