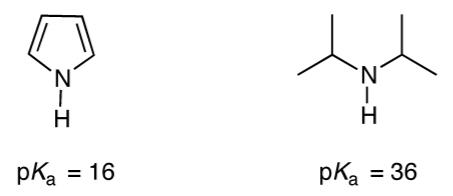
Pyrrole is much more acidic than diisopropylamine. Explain. pka = 16 pKa %3D pka = 36 Z-I
Fantastic news! We've Found the answer you've been seeking!
Question:
Pyrrole is much more acidic than diisopropylamine. Explain.
Transcribed Image Text:
pka = 16 pKa %3D pka = 36 Z-I Z-エ pka = 16 pKa %3D pka = 36 Z-I Z-エ
Expert Answer:
Answer rating: 100% (QA)
This can be explained on the basis of the protonic concept ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Pyrrole is much more acidic than a typical secondary amine (pKa's of 16 and 40, respectively). With the aid of diagrams, explain this difference. pyridine yrrole ZI
-
One of the following hydrocarbons is much more acidic than the others. Indicate which one, and explain why it is unusually acidic.
-
The O ? H hydrogen in acetic acid is much more acidic than any of the C ? H hydrogens. Explain this result using resonance structures. Acetic acid
-
Day and Night formed an accounting partnership in 2014. Capital transactions for Day and Night during 2014 are as follows: Partnership net income for the year ended December 31, 2014; is $68,400...
-
A 2.500-mol sample of phosphorus pentachloride, PCl5, dissociates at 160oC and 1.00 atm to give 0.338 mol of phosphorus trichloride, PCl3, at equilibrium. What is the composition of the final...
-
When a borrower pledges certain properties of value as a guarantee that she will pay the debt back, the creditor is called a _____ party. a. Lender b. Secured c. Collateral d. Indebted
-
The Wide World of Fluids article titled "Bow Thrusters,".The bow thruster on the boat shown in Fig. P5.77 is used to turn the boat. The thruster produces a \(1-\mathrm{m}\)-diameter jet of water with...
-
The XYZ Corporation was duly organized on July 10. Its certificate of incorporation provides for total authorized capital of $1 million, consisting of ten thousand shares of common stock with a par...
-
If a 1000 taka face value bond paying 9% coupon semi-annually when the interest rate is 16% but selling in the market for 680 taka. Should you buy this bond? Find Current Yield and YTM of this bond.
-
The operations manager of a firm that produces frozen dinners had received numerous complaints from supermarkets about the firms Chick-n-Gravy dinners. The manager then asked her assistant, Ann, to...
-
Does a matrix or customer-oriented structure lend itself better to forming joint ventures and alliances? please explain.
-
A company has the following results for the four years to 31 March 2021: Calculate the total repayment of corporation tax (with interest) to which the company is entitled, assuming that: (a) all...
-
In 2013, Shaun bought 20% of the ordinary shares of an unlisted trading company. The shares cost 140,000. He owned the shares until January 2021 when he gave all the shares to a friend. On the date...
-
Calculate the CGT payable in relation to each of the following disposals, assuming in each case that the annual exemption is fully utilised against other gains, that there are no allowable losses and...
-
Timberlake Ltd prepares accounts to 31 March each year. The company made the following disposals of chargeable assets in the year to 31 March 2021: (i) In February 2021, a rare Bentley motor car used...
-
Stephen is a UK resident taxpayer with two different sources of income. He works part time as an IT consultant for a small number of clients, on a project management basis. A separate contract is...
-
down? How far from the end of the ramp does the skateboarder touch down? Express your answer with the appropriate units. View Available Hint(s) A ? l = Value Units Submit Previous Answers Units...
-
To balance the chemical equation SiH3 + O2 SiO2 + HO, you could introduce coefficients a, b, c, d and write aSiH3 + bO2 cSiO + dHO then write linear equations for each element. The equation for Si...
-
When ethyl bromide is added to potassium tert-butoxide, the product is ethyl tert-butyl ether. (a) What happens to the reaction rate if the concentration of ethyl bromide is doubled? (b) What happens...
-
(A true story.) While organizing the undergraduate stockroom, a new chemistry professor found a half-gallon jug containing a cloudy liquid (bp 100105 C), marked only STUDENT PREP. She ran a quick...
-
Give both the IUPAC name and the common name for each alcohol. (a) CH3CH2CH(OH)CH3 (d) (CH3)2CHCH2CH2OH OH
-
In the second quarter of 2021, personal consumption expenditures, exports, and imports increased. Investment and government expenditure decreased. Real GDP increased by 6.5 percent following a 6.3...
-
When real GDP increased in the second quarter of 2021, consumption expenditure, exports, and imports increased. Fixed investment decreased, which included a decrease in business inventory investment....
-
Real exports of goods and services increased 6 percent in the second quarter of 2021, compared with a decrease of 2.9 percent in the first quarter of 2021. Real imports of goods and services...

Study smarter with the SolutionInn App


