Question: The overhead system for a distillation column is shown in Figure. The composition of the total distillates is indicated, with 10 mol% of it being
The overhead system for a distillation column is shown in Figure. The composition of the total distillates is indicated, with 10 mol% of it being taken as vapor. Determine the pressure in the reflux drum, if the temperature is 100?F. Use the following K-values by assuming that K is inversely proportional topressure.
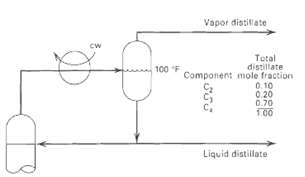
Vapor distitlate cw Total distillate Component mole fraction 0. 10 0.20 0.70 100 100 F Liquid distillate
Step by Step Solution
3.55 Rating (172 Votes )
There are 3 Steps involved in it
As shown in Exercise 424 the compositions of net equi... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
37-E-C-E-S-P (117).docx
120 KBs Word File


