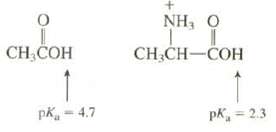
The pKa of the carboxylic acid group of acetic acid is 4.7. The pKa of the carboxylic
Question:
The pKa of the carboxylic acid group of acetic acid is 4.7. The pKa of the carboxylic acid group of the conjugate acid of the amino acid alanine is 2.3. Explain the difference in these pKa values.
Transcribed Image Text:
CH,COH 1 pk = 4.7 NH, Ο I CH₂CH-COH pk = 2.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
The NH 3 is an inductiv...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In glycine, the carboxylic acid group has Ka = 4.3 X 10-3 and the amino group has Kb = 6.0 X 10-5. Use these equilibrium constant values to calculate the equilibrium constants for the following. a....
-
Acid HA has pKa = 20; acid HB has pKa = 10. (a) Which is the stronger acid? (b) Will an acid-base reaction with an equilibrium lying to the right take place if Na+A- s added to HB? Explain your...
-
Acetic acid is continuously extracted from a 3 wt% dilute solution in water with a solvent of isopropyl ether in a mixer-settler unit. The flow rates of the feed and solvent are 12,400 and 24,000...
-
Simplify the expression 2x + (x + 1) into a single x + 1 fraction. The numerator of your answer is: The denominator of your answer is:
-
Describe the factors that managers may consider when assessing the significance of a variance. Why do we need to assess the significance of variance?
-
Purple Feet Wine, Inc., receives an average of $19,000 in checks per day. The delay in clearing is typically three days. The current interest rate is .019 percent per day. a. What is the company's...
-
What is the relationship of technology to the information system architecture?
-
What is the net asset value of an investment company with $10,000,000 in assets, $790,000 in current liabilities, and 1,200,000 shares outstanding?
-
Suppose a 20-year Treasury bond with 5% semiannual coupon is currently traded at a yield of 6% (APR). 1) Is the price above or below 100? 2) What is the current price of the bond? 3) If the yield...
-
What could Peter and Matts manager do to help them resolve their conflict? The case suggests that there is research to support the notion that avoiding conflict stifles creativity. Is there such...
-
Explain which nitrogen in the ring of the amino acid histidine is the stronger base: N- N-H CH + HN CHCO, Histidine
-
There are two isomeric conjugate acids that produce the following base. Show the structure of each and explain how they produce the same base upon loss of a proton. :0: CH3-C=CH
-
A bank manager has been presented with a new brochure that was designed to be more effective in attracting current customers to a personal financial counseling session that would include an analysis...
-
Write a program Checkerboard that takes an integer command-line argument n and uses a loop nested within a loop to print out a two-dimensional n-by-n checkerboard pattern with alternating spaces and...
-
What are the three typical levels of firm strategy?
-
What other solutions could you see be effective at improving employee motivation and reducing the turnover rate? Why do you believe these solutions would be useful?
-
How else might the manager have handled the situation to prevent potential issues, including a negative impact on the teams performance?
-
What would be the most effective option to increase employee motivation to stay and reduce the driver turnover rate? Why do you believe this option will be effective?
-
"What were they smoking?" The Fortune cover story featured those words in a 3.5-inch headline, as well as photos of Chuck Prince, Citigroup (\$9.8 billion loss), Jimmy Cayne, Bear Stearns ( \(\$...
-
What is a manufacturing system?
-
If |x 4| < 0.1 and |y 7| < 0.2, use the Triangle Inequality to estimate |(x + y) 11|.
-
How would you prepare 1-phenyl-2-butanone, C6H5CH2COCH2CH3, from benzyl bromide, C6H5CH2Br? More than one step is required.
-
Cyclopentanecarboxylic acid and 4-hydroxycyclohexanone have the same formula (C6H10O2), and both contain an OH and C = O group. How could you distinguish between them by IR spectroscopy?
-
How could you distinguish between the isomers Cyclopentanecarboxylic acid and 4-hydroxycyclohexanone by 1H and 13C NMR spectroscopy?
-
To set up an optimal portfolio problem considering transactions costs, we need to adjust the expected return of stocks and calculate the transactions-cost-adjusted expected return. Suppose that a $1...
-
How to calculate the average precision (AP) for each adopted query and the mean average precision (MAP), based on both arithmetic and geometric means, must be calculated in terms of interpolated...
-
Using the relative contribution of the B to the overall expected portfolio return, show whether index leverage or single-stock leverage results in better leverage for a portfolio manager who is good...

Study smarter with the SolutionInn App


