There are two isomeric conjugate acids that produce the following base. Show the structure of each and
Question:
There are two isomeric conjugate acids that produce the following base. Show the structure of each and explain how they produce the same base upon loss of a proton.
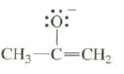
Transcribed Image Text:
:0: CH3-C=CH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
The anion has two resonance stru...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
(a) Describe cash equivalents and explain how they are reported. (b) How should restricted cash funds be reported on the balance sheet?
-
Identify two types of value streams and explain how they differ.
-
Name two types of organizational design and explain how they differ.
-
what is resourced based view in public administration and why is it important?
-
What might an unfavourable direct labour efficiency variance indicate? What actions could be taken to correct this variance?
-
White Whale Corporation has an average daily cash balance of $1,300. Total cash needed for the year is $43,000. The interest rate is 5 percent, and replenishing the cash costs $8 each time. What are...
-
How and why does the architecture of the information system differ from software architecture?
-
In a 3M Privacy Filters poll, 806 adults were asked to identify their favorite seat when they fly, and 492 of them chose a window seat. Use a 0.01 significance level to test the claim that the...
-
Alexandra bought a 7-year bond at a 7.50% discount to its par value of $1,000 three years ago. The bond has a 4.70% coupon rate payable semiannually. What was her effective annual rate of return if...
-
The following bill of materials represents the major components for a computer system. Complete the MRP records below. Note the following: Production plans (the MPS) for the 800 Deluxe computer...
-
The pKa of the carboxylic acid group of acetic acid is 4.7. The pKa of the carboxylic acid group of the conjugate acid of the amino acid alanine is 2.3. Explain the difference in these pKa values....
-
When a is reacted with hydroxide ion, isomer b is formed. (a) Explain why b is favored over a at equilibrium. (b) Show the structure of the conjugate base of a and explain how this isomerization...
-
Many matings are observed in a species of bird. Both female and male birds come in three colors: red, blue, and green. For each experiment, find the marginal distributions for both sexes and the...
-
In what ways do you believe providing special work arrangements or accommodations for employees impacts employee motivation? How does it help? How does it hurt?
-
What circumstances might cause a group to perform worse than its best individual?
-
What can groups do to make group decision making more effective?
-
What are the shortcomings of the trait approach in psychological criminology?
-
Claudia and Albert increased their net worth from \($250,000\) to \($270,000\) during the current year. Their combined income was \($60,000\) and the only changes to their assets was an increase in...
-
Enron Corp. was an energy company that was incorporated in Oregon in 1985, with its principal executive offices located in Houston, Texas. By the end of 2001, Enron Corp. was the world's largest...
-
In the operation of an automated production line with storage buffers, what does it mean if a buffer is nearly always empty or nearly always full?
-
Solve |3x + 2| 4.
-
Give IUPAC name for the following carboxylic acids (reddish brown = Br). (a) (b) (d) (c)
-
Would you expect the following carboxylic acids to be more acidic or less acidic than benzoic acid? Explain. (Reddish brown = Br) (a) (b) (d) (c)
-
The following carboxylic acid can?t be prepared from an alkyl halide by either the nitrile hydrolysis route or the Grignard carboxylation route. Explain.
-
Can social norms be understood as emergent properties of social interactions, reflecting negotiated agreements among members of a society regarding acceptable behavior and moral standards?
-
A quantitative portfolio manager is very good at managing against the Russell 2000 Index. In fact, she is expected to achieve an B of 0.5% per month. Unfortunately, her benchmark is the S&P 500. She...
-
Suppose that a QEPM department hires a new quantitative equity portfolio manager to manage one of its portfolios. Suppose that the portfolio managers benchmark is the S&P 500, with a Sharpe ratio of...

Study smarter with the SolutionInn App


