Upon consideration of the SiO2???Al2O3 phase diagram, Figure, for each pair of the following list of compositions,
Question:
Upon consideration of the SiO2???Al2O3 phase diagram, Figure, for each pair of the following list of compositions, which would you judge to be the more desirable refractory? Justify your choices.
(a) 20 wt% Al2O3-80 wt% SiO2 and 25 wt% Al2O3-75 wt% SiO2
(b) 70 wt% Al2O3-30 wt% SiO2 and 80 wt% Al2O3-20 wt%SiO2
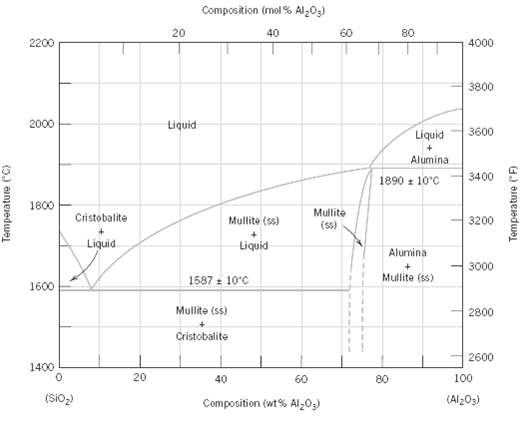
Transcribed Image Text:
Composition (mol % Al203) 80 20 40 60 2200 4000 3800 2000 Liquid Liquid 3600 Alumina 3400 1890 + 10°C 1800 Mullite (s) Cristobalite 3200 Mullite (ss) Liquid Liquid Alumina 3000 Mullite (ss) 1587 : 10°C 1600 Mulite (ss) 2800 Cristobalite 2600 1400 20 100 40 60 80 (SIO,) (Al203) Composition (wt % Al;03) Temperature ("C) Temperature ("F)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
a The 25 wt Al 2 O 3 75 wt SiO 2 will be more desirable because ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
For each pair of the following molecules, indicate whether its members are identical, structural isomers, conformers, or stereoisomers. How would you describe the relation between conformations when...
-
Upon consideration of the SiO2-Al2O3 phase diagram, Figure 12.27, for each pair of the following list of compositions, which would you judge to be the more desirable refractory? Justify your choices....
-
For each pair of the following compounds identify which compound would react more rapidly in an E1 reaction. a. b. CI .CI CI CI
-
Marsden Corp has developed a new strategic plan after rushing defective products to market hurt the company's sales and image. The strategic plan's initiatives are to focus on quality and develop...
-
Briefly describe the Religion of the Iceland. Briefly describe the Government of the Iceland. Briefly describe the Technology of the Iceland. How do these cultural elements described in this peer...
-
Dr. Helen Samoa opened a medical practice specializing in physical therapy. During the first month of operation (December), the business, titled Dr. Helen Samoa, Professional Corporation (P.C.),...
-
What are the three types of employees who work in MNEs? Explain how an HCN could change to become a TCN in an MNE.
-
Identify the best answer for each of the following: 1. On July 1, 20X5, Lilydale Hospitals board of trustees designated $200,000 for expansion of outpatient facilities. The $200,000 is expected to be...
-
Head First Company plans to sell 5, 100 bicycle helmets at $78 each in the coming year Product costs include Direct materials per helmet $32 Direct labor per helmet 5.50 Variable factory overhead per...
-
1. How would you describe the current charity industry structure? How could it change if consolidation increases and what would be the benefits and disadvantages? 2. Which of Porters five forces are...
-
Find the maximum temperature to which the following two magnesia???alumina refractory materials may be heated before a liquid phase will appear. (a) A spinel-bonded alumina material of composition 95...
-
Compute the mass fractions of liquid in the following refractory materials at 1600C (2910F): (a) 6 wt% Al2O3-94 wt% SiO2 (b) 10 wt% Al2O3-90 wt% SiO2 (c) 30 wt% Al2O3-70 wt% SiO2 (d) 80 wt% Al2O3-20...
-
The function f is defined by f: x 7 5 cos 2x for 0 x . The function g is defined by g: x x + /2 for 0 x . a) State the range of f. b) Sketch the graph of y = f(x). c) Find the function fg.
-
The cantilever beam in Figure P14.9 is connected to a bar at joint 2 by a pin. Compute all reactions. Given: \(E=30,000 \mathrm{kips} / \mathrm{in}\). \(^{2}\). Ignore axial deformation of the beam....
-
List four macro-segmentation variables and explain their value in business markets.
-
The ways in which people from different countries buy cups of tea or coffee have been steeped in tradition. The English tearoom has been quintessentially English, usually providing homely...
-
What is the standard of care used to judge health care professionals?
-
A quick scan of any business telephone directory or an Internet search would reveal an interesting trend occurring in the marketing of service businesses. It is the idea of service companies...
-
In what ways could you enhance your relationship strategy with prospective and existing customers by effectively using social media?
-
Suppose you are comparing just two means. Among the possible statistics you could use is the difference in means, the MAD, or the max min (the difference between the largest mean and the smallest...
-
Sketch a graph of the potential energy of two atoms as a function of the distance between them. On your graph, indicate how bond energy and bond distance are defined.
-
What is the difference between a coherent precipitate and a distinct second-phase particle?
-
What is overaging?
-
What types of heating and cooling conditions are imposed in an I-T or T-T-T diagram? Are they realistic for the processing of commercial items?
-
E3-20 (Algo) Analyzing, Recording, and Evaluating the Effects of Income Statement Transactions [LO 3-3, LO 3-4, LO 3-5] [The following information applies to the questions displayed below.] MyBnB...
-
Required information [The following information applies to the questions displayed below.] The following data reports on the July production activities of the Molding department at Ash Company....
-
Question 1 Vieux Enterprise's total overhead costs at various levels of activity follow: Month Machine- Hours Total Overhead Costs April 70,000 $198,000 May 100,000 270,000 June 80,000 222,000 July...

Study smarter with the SolutionInn App


