Question: Using known reactions and mechanisms discussed in the text, complete the reactions given in Fig. P19.46 on p. 942. Fig. P19.46 NaBH4 CH OH
Using known reactions and mechanisms discussed in the text, complete the reactions given in Fig. P19.46 on p. 942.
Fig. P19.46
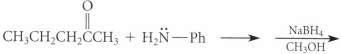
NaBH4 CH OH
Step by Step Solution
3.37 Rating (166 Votes )
There are 3 Steps involved in it
An initially formed imi... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
902-O-EE (155).docx
120 KBs Word File


