Question: Using the data in Table 5.2, compute the value of D for the diffusion of zinc in copper at 650?C. Activation Energy Qa Calculated Values
Using the data in Table 5.2, compute the value of D for the diffusion of zinc in copper at 650?C.
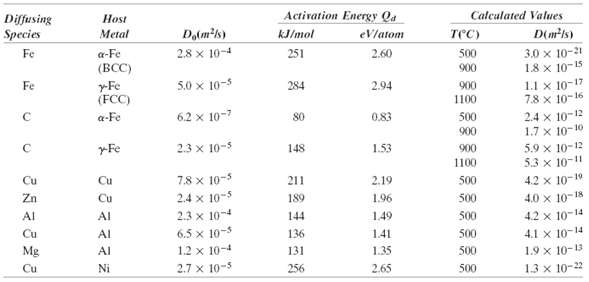
Activation Energy Qa Calculated Values Diffusing Species Host Do(mk) (mts) Metal kJ/mol ) eVlatom 3.0 x 10-21 1.8 x 10-15 1.1 x 10-17 7.8 x 10-16 a-Fe (BCC) Fe 2.8 x 10-4 251 2.60 500 900 5.0 x 10-5 Fe y-Fe (FCC) 284 2.94 900 1100 2.4 x 10-12 1.7 x 10-10 6.2 x 10-7 a-Fe 80 0.83 500 900 5.9 x 10-12 5.3 x 10-11 yFe 2.3 x 10-5 1.53 148 900 1100 7.8 x 10-5 Cu 4.2 x 10-19 Cu 211 2.19 500 Zn 2.4 x 10-5 500 4.0 x 10-18 Cu 189 1.96 2.3 x 104 1.49 500 4.2 x 10-14 Al AI 144 6.5 x 10-5 1.2 x 10-4 4.1 x 10-14 Cu AI 136 141 500 1.9 x 10-13 1.3 x 10-22 Mg Al 131 1.35 500 2.7 x 10-5 Cu Ni 256 2.65 500
Step by Step Solution
3.40 Rating (175 Votes )
There are 3 Steps involved in it
Incorporating the appropriate data from Table 52 into Equation 58 leads to 1 In the lefthand window ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
33-E-M-S-E-M-S (142).docx
120 KBs Word File


