We?ve seen that the Diels?Alder cyclo addition reaction is a one-step, pericyclic process that occurs through a
Question:
We?ve seen that the Diels?Alder cyclo addition reaction is a one-step, pericyclic process that occurs through a cyclic transition state. Propose a mechanism for the following reaction:
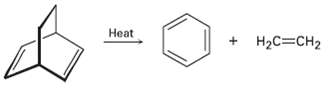
Transcribed Image Text:
Нeat Нeat + H2C=CH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
DielsAlder reaction are reversible when the produ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the following reaction (remember to use curved arrows when showing a mechanism): CH3CHCH-CH-OH CH,C-CH, CH,CHCH-CH-OCCH, + CH CH
-
Propose a mechanism for the following reaction that explains why the configuration of the asymmetric carbon in the reactant is retained in the product: COO NaNO2 HCl NH2 COO
-
Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the canvas and connecting them with bonds. Add charges where needed. Electron flow arrows should start on the...
-
The following list shows the top six pharmaceutical companies in the United States and their sales figures ($ millions) for a recent year. Use this information to construct a pie chart and a bar...
-
What are the pros and cons of offering sick leave benefit?
-
* Determine i0 (t) in the network shown in Fig. 16.48. 4 2 H 4
-
The following stream at \(100^{\circ} \mathrm{F}\) and 250 psia is to be separated into the four indicated products. Also, given is the cost of each of the unique separators. Determine: (a) The best...
-
An examination of Frosty Corporations records reveals the following transactions: a. On December 31, the physical inventory of raw material was 9,950 gallons. The book quantity, using the moving...
-
Suppose you write a June expiration call option on Tesla with exercise price $85 and at the same time, write a June Tesla put option with exercise price $80. The premium of the call option is $0.90...
-
Presented below is an aging schedule for Bryan Company at December 31, 2021. At December 31, 2021, the unadjusted balance in Allowance for Doubtful Accounts is a credit of $8,000. Instructions a....
-
Norbornadiene (Problem 14.41) can be prepared by reaction of chloroethylene with 1, 3-cyclopentadiene, followed by treatment of the product with sodium ethoxide. Write the overall scheme, and...
-
In light of your answer to Problem 14.43, propose a mechanism for the followingreaction: CH CH + CO2 Heat "CCH do,c, H a-Pyrone CHC-
-
Calculate the surface area of (a) A 1 g single crystal of cristobalite (density = 2.3 g/cm 3 , assume the crystal takes a spherical shape), (b) A uniform powder of spherical crystals each with a...
-
generate an EMAIL to your mailing list, to promote Your blog post about sleep and mental health and ask them to learn more with a link to the blog
-
6. (2pt) Michael, a 10-year-old boy in generally good health, went to a birthday party on Sunday with his family. He ate many different foods but was the only one in the family to eat the undercooked...
-
= How would you prepare 1.0 litres of 1000 ppm lead standard in 2% nitric acid, starting from lead acetate (Pb(C2H3O2)2; molecular weight 325.28) and concentrated nitric acid (100%)? Show your...
-
How can you use Kotter's 8-Step Process for organizational change for a future career?
-
Swifty Company purchased factory equipment for $ 6 4 2 0 0 0 . Useful hours for the machine are estimated to be 1 7 7 0 0 0 . The estimated salvage value is $ 5 2 0 0 0 , and the estimated useful...
-
Mark glanced up at the clock on his office wall. It read 2:30 P.M. He had scheduled a 3:00 P.M. meeting with George Hang-ten Baldwin, chief executive officer of Surfer Dude Duds, Inc. Surfer Dude...
-
For the vector whose polar components are (Vr = 1, Vθ = 0), compute in polars all components of the second covariant derivative Vα;μ;ν. To find...
-
A solution contains 3.95 g of carbon disulfide (CS 2 ) and 2.43 g of acetone (CH 3 COCH 3 ). At 35 C the vapor pressures of pure carbon disulfide and pure acetone are 515 torr and 332 torr,...
-
Predict the major products of the following reactions: (a) (b) (c) (d) Zn(Hg) HCI, H20 (1) H2NNH (2) KOH, heat (1) N2H4 2) KOH, heat O HCI, H,O
-
Use equations to show the fragmentation leading to each numbered peak in the mass spectrum of octan-2-one. 100 80 e 60 2 40 20 0 43 85 113 12 10 20 30 40 50 60 70 80 90 100 110 120 130 40 150 160
-
The following compounds undergo McLafferty rearrangement in the mass spectrometer. Predict the masses of the resulting charged fragments. (a) Pentanal (b) 3-methylhexan-2-one (c) 4-methylhexan-2-one
-
Let R denote the region in the xy-plane that lies in the first quadrant and is bounded by the lines y = 1 - x, y = 2 - x, y = x + 1, and y = x - 1. Set u=x+y and v = y-x. (a) Solve for x and y in...
-
Reply to The culture established by Mr. Kelleher places importance on employee happiness, which directly influences customer satisfaction and overall organizational achievements. To maintain this...
-
First, technology has a major impact on talent shortage. With the increased use of AI, there are fewer data analysis jobs required, so technology can potentially replace some forms of supply chain...

Study smarter with the SolutionInn App


