Question: In light of your answer to Problem 14.43, propose a mechanism for the followingreaction: CH CH + CO2 Heat CCH do,c, H a-Pyrone CHC-
In light of your answer to Problem 14.43, propose a mechanism for the followingreaction:
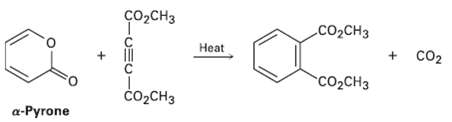
CH CH + CO2 Heat "CCH do,c, H a-Pyrone CHC-
Step by Step Solution
3.28 Rating (157 Votes )
There are 3 Steps involved in it
A DielsAlder reaction between opyrone diene and the alkyne dienophile yields t... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-S (284).docx
120 KBs Word File


