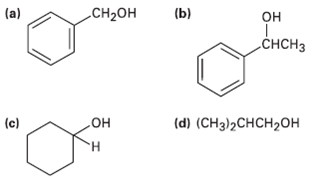
What carbonyl compounds give the following alcohols on reduction with LiAlH4? Show allpossibilities. (a) CH- (b)
Question:
What carbonyl compounds give the following alcohols on reduction with LiAlH4? Show allpossibilities.
Transcribed Image Text:
(a) CH-он (b) он „CHCH3 (d) (CHз)2СHCH20Н (c) но
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
i OH obotowor a CCH3 1 LIAIH4 2H30 d Benzyl alcohol may b...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show what alcohols and carbonyl compounds give the following derivatives. (a) (b) (c) (d) (e) (f) CH CH,O OCH,CH CH O-CH CH3 CH3 CH-C H O-CH CH a,0 OX
-
Show what amines and carbonyl compounds combine to give the following derivatives. (a) (b) (c) (d) (e) (f) Ph-CH=N-NH-C-NH, NOH N NHPh HNN : O
-
What carbonyl compounds are required to prepare a compound with molecular formula C10H10Owhose spectrum is shown? (ppm)
-
On 1 June 1998, Alice bought a house in Derby for 45,000. - She occupied the house as her PPR until 1 May 2000 when she left to work in Exeter, living in rented accommodation. - She returned to the...
-
Illustrate a policy, an objective, and a functional tactic in your personal career strategy.
-
Answer Problem 5.32 for values 2.0 times the upper limit of normal? Blood Chemistry In pharmacologic research a variety of clinical chemistry measurements are routinely monitored closely for evidence...
-
In Example 3.1, we see that \(27.5 \%\) of US adults are college graduates. (a) Use StatKey or other technology to generate a sampling distribution for the sample proportion of college graduates...
-
Ghana is a producer and exporter of crude oil. Since Ghana is a relatively small crude-oil-producing country, its actions do not affect world prices; as an exporter, Ghana faces a foreign demand...
-
7. A bullet with a mass of 0.003kg is fired into a wooden block that lies on the top of a frictionless horizontal table. The bullet becomes embedded after the collision with the block of mass 0.7kg...
-
You have completed your As-Is diagram Assignment: Generate a list of ideas for the new process (brainstormed) Select the top 2 most promising ideas and assess them in context by defining its impact...
-
What reagent would you use to accomplish each of the following reactions? , 2. (a) CHCH-CH2co CHH2CH2cH , 2. (b) CHH2H2H2 CHH2H2cH (c)
-
Show the products obtained from addition of methyl magnesium bromide to the following compounds: (a) Cyclopentanone (b) Benzophenone (diphenyl ketone) (c) 3-Hexanone
-
Alice, Bill, and Charles each received an equal number of shares when they formed King Corporation a number of years ago. King has used the cash method of accounting since its inception. Alice, Bill,...
-
Stoney Corporation invest $6,500 each in Projects X and Y. Cash flows from these investment are listed below Investment X Investment Y Year 1 $1000 $1300 Year 2 1800 2000 Year 3 1700 1100 Year 4 2000...
-
Tony Thomas has been found to be in contempt of a Florida Family Court order. What enforcement tools are available to the judge? Explain by discussing the facts and ruling of a Florida family law...
-
Why would a country decide to franchise abroad? Explain
-
1. Determine the Boolean expression in sum-of-products form for the following truth table. Do not simplify your answer. A B C D 0 0 0 1 0 0 1 1 0 1 0 1 0 1 1 0 1 0 0 1 1 0 1 1 1 1 1 1 0 1 0 0 2. (0.5...
-
An 86 kg passenger in an SUV traveling at 1200 km/h is wearing a seat belt. The driver slams on the brakes and the SUV stops in 42 m. Find the magnitude of the force exerted by the seat belt on the...
-
What factors do you think a defense attorney considers in deciding whether to advise a client to testify in his or her own behalf at trial?
-
What types of inventory issues Starbucks might reflect upon at the end of each year? The mission of Starbucks is to inspire and nurture the human spiritone person, one cup, and one neighborhood at a...
-
Construct a concept map using the ideas of packing of spheres and the structure of metal and ionic crystals.
-
Starting with appropriate unlabeled organic compounds, show syntheses of each of the following: (a) C6H5-C¡C-T (b) (c) CH3CH2CH2OD CH3
-
(a) Arrange the following compounds in order of decreasing acidity and explain your answer: CH3CH2NH2, CH3CH2OH, and CH3CH2CH3. (b) Arrange the conjugate bases of the acids given in part (a) in order...
-
Arrange the following compounds in order of decreasing acidity: (a) CH3CH==CH2, CH3CH2CH3, CH3CCH (b) CH3CH2CH2OH, CH3CH2CO2H, CH3CHClCO2H (c)
-
Complete the table by: i. Defining each type of taxable transaction data provided below. ii. Listing one unique source of information for each type of taxable transaction data listed below. iii....
-
You are a partner in a CPA firm. You have a client that is being audited by the IRS. The IRS is using an indirect method of proof of his income. Your client is an attorney but not a tax attorney and...
-
Duncan Corp enters into a contract with Perry Company to sell four different products and each of the products is considered a separate performance obligation. The total price of the contract is...

Study smarter with the SolutionInn App


