What reagent would you use to accomplish each of the following reactions? , 2. (a) CHCH-CH2co
Question:
What reagent would you use to accomplish each of the following reactions?
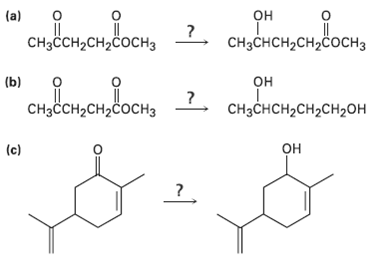
Transcribed Image Text:
обенодо, 2. ореородвонть (a) CHзснCH-CH2coснз CHзссH2CH2cосHз сононвоси, 2. он (b) CHзснсH2сH2сH2он CHзҫсH2сH2cосHз он (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
1 NaBH4 2 H30 OH CH3CHCHCHCOCH 3 NaBH4 reduces aldehydes and k...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What reagents would you use to accomplish a substitution with retention of configuration; for example? OH SH (R)-2-Butanol (R)-2-Butanethiol
-
Identify the reagent you would use to accomplish each of the following transformations: (a) Cyclobutanol bromocyclobutane (b) tert-Butanol tert-butyl chloride (c) Ethyl chloride ethanol
-
Each of the following reactions has been reported in the chemical literature and proceeds cleanly in good yield. Identify the principal organic product in each case. (a) (b) (c) (d) (e) (f) (g) (h)...
-
Alistair bought a house on 1 April 2000 for 125,000 and occupied the entire house as his principal private residence until 1 November 2008. As from that date, he rented out two rooms (comprising...
-
Why are short-term objectives needed when long-term objectives are already available?
-
The Metropolitan Museum of Art in New York City is one of the best in the world. Under an 1893 law, it must admit the public free of charge five days and two evenings a week. The Met has a long...
-
Gender in the Rock and Roll Hall of Fame From its founding through 2015, the Rock and Roll Hall of Fame has inducted 303 groups or individuals. Forty-seven of the inductees have been female or have...
-
Matt owns common shares of Maeb Inc. The adjusted cost base and paid-up capital of these shares is $300,000 and the FMV is $900,000. In the course of a capital reorganization, the following two...
-
Sales Variable expenses $ 98,000 100% 39, 200 40% Contribution margin 58,800 60% 45,080 Fixed expenses Net operating income $ 13,720 Required: 1. What is the company's degree of operating leverage?...
-
Pear Corporation acquired 75 percent ownership of Sugar Company on January 1, 20X1, at underlying book value. At that date, the fair value of the noncontrolling interest was equal to 25 percent of...
-
Predict the products of the following reactions: CH (a) 1. CHCH2- 2. NaOH, H2O2 "CH (b) 1. HglOAc)2. 0 2. NABH4 (c) CCH2CH2CH2 CH2CH2CH2CH3 C=C 1. Os04 2. NaHSO3, H20 -
-
What carbonyl compounds give the following alcohols on reduction with LiAlH4? Show allpossibilities. (a) CH- (b) CHCH3 (d) (CH)2HCH20 (c)
-
The McCracken County Humane Society (MCHS), which is part of a county's reporting entity, established a permanent fund to provide support for its pet neutering program. As of the start of the year,...
-
Using your textbook and lecture notes as guides, discuss if it is possible to be a great leader of employees in a highly mechanistic organization. What special talents or abilities might be required?
-
Ralley Corporation sells a single product at a price of $ 2 7 5 per unit. Variable cost per unit is $ 1 3 5 and fixed costs total $ 3 5 6 , 8 6 0 . If sales are expected to be $ 8 2 5 , 0 0 0 , what...
-
Describe how you go about starting a small business and what resources are needed.
-
.Why is value chain analysis important from a strategic planning perspective?
-
What are the financial functions carried out by financial markets in an effective financial system? Explain
-
In England, the voir dire process is conducted by the trial judge and is extremely limited. Do you think the American system of criminal justice would have more credibility if the voir dire were...
-
Explain the term "Equivalent Units". Why are they calculated in process costing? [4 Marks] [minimum 350 words]
-
Construct a concept map showing the ideas contained in a phase diagram.
-
What reaction will take place if ethyl alcohol is added to a solution of HCC:- Na+ in liquid ammonia?
-
Acid HA has pKa = 20; acid HB has pKa = 10. (a) Which is the stronger acid? (b) Will an acid-base reaction with an equilibrium lying to the right take place if Na+A- s added to HB? Explain your...
-
Which of the following are potential Lewis acids and which are potential Lewis bases? (a) (b) (c) (C6H5)3P: (d) (e) (CH3)3B (f) H: - CH.CH-N-CH CH3 CH3 H3C-C CH Br:
-
Problem 2 [55 marks] Consider a thin solid disk of radius a and material properties, E and v, which is placed inside a disk with a hole of the inner radius b = a and the outer radius a with materials...
-
(1) The volume of the cylinder was 1L at 50 C. What will be the new volume Vat T=100 C? Assume that the mass on top of the cylinder is kept the same. (Hint: P1=P2) P=const n=const V2 T2 = V V Ti (2)...
-
8. Mr. McCloskey is driving his truck along a straight road when he notices a small dark object 500 ft ahead. He steps on the brakes, then recognizes that the object is a duck standing in the middle...

Study smarter with the SolutionInn App


