What carbonyl compounds would you reduce to prepare the following alcohols? List all possibilities. (b)
Question:
What carbonyl compounds would you reduce to prepare the following alcohols? List all possibilities.
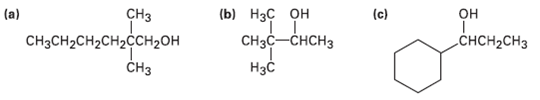
Transcribed Image Text:
(b) Нзс он Cнзс—снсHз Нас |(a) (c) СHз CHзCH2CH2сH2ссH2он CНз он CHCH-CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
a b c Alcohol CH3 CH3CHCHCHCCHOH ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What alkenes might be used to prepare the following alcohols by hydroboration/oxidation? (a) CH3 (b) CHH (c) .CH- CHCHCH2CH2OH
-
What reagents would you use to prepare the following compounds? a. b. CH CCH CH:C 0 CH3CCH-CH-CH (COCH-CH3 )2
-
What alkyl halides would you use to prepare the following ketones by an acetoacetic ester synthesis? CH (a) (b) CHH,H,H3 -CH-CH2CH2CCH
-
Chambers Corporation purchased a piece of equipment for $36,000. It estimated a 6-year life and $6,000 salvage value. Thus, straight-line depreciation was $5,000 per year [($36,000 $6,000) 6]. At...
-
Describe SWOT analysis as a way to guide internal analysis. How does this approach reflect the basic strategic management process?
-
Fit a regression line relating 5-year lung-cancer mortality to annual cigarette consumption when each is expressed in the log 10 scale? Cancer The following statistics are taken from an article by...
-
If each point of the sample space of Figure 3.12 represents an outcome having the probability find, 32
-
You and your friends are doing physics experiments on a frozen pond that serves as a frictionless, horizontal surface. Sam, with mass 80.0 kg, is given a push and slides eastward. Abigail, with mass...
-
What are the types of non-equity entry modes?
-
1. Imagine that you run the central bank in a large open economy with a floating exchange rate. Your goal is to stabilize income, and you adjust the money supply accordingly. Under your policy, what...
-
What Grignard reagent and what carbonyl compound might you start with to prepare the following alcohols? CH (a) (b) (c) CHCH-CHCH2CH CHH2H H2c=C "CH- (d) (e) (f) .CH- "
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
Jolene receives tax-exempt interest of $33,000 on bonds that are classified as private activity bonds. She properly excludes the $33,000 from her gross income for regular income tax purposes. Jolene...
-
Understanding Civil Law in ONTARIO Canada Civil Law covers a lot of areas of Law. For this please highlight 4 separate areas of Civil Law for ONTARIO Canada. (Tort Law, Family Law, Employment Law...
-
What are the possible roles that an Marriage and Family Therapist can play during divorce or custody proceedings? understandings of the Collaborative Divorce approach and the role of a mental health...
-
Prepare journal entries for the following transactions of Nutrition Consulting Inc. a. b. Nov. 5 Paid $5,000 cash for website maintenance services covering a 3-month period, beginning December 1. C....
-
Howard Company has the following information for the current period for the Welding Department: Costs of beginning work-in-process: Direct materials Conversion costs Costs added during the period:...
-
Question Little Kindnesses, Inc. is a tax-exempt charity organized as a corporation. It has unrelated business taxable income of $30,000. How much tax is due on the unrelated business taxable income?
-
In 1987 Douglas Deville was convicted in a state court for felonious possession of cocaine. He received probation for that offense. Three years later, Deville was found guilty of another drug-related...
-
In the figure, two loudspeakers, separated by a distance of d1 = 2.63 m, are in phase. Assume the amplitudes of the sound from the speakers are approximately the same at the position of a listener,...
-
In a capillary rise experiment, the height (h) to which a liquid rises depends on the density (d) and surface tension () of the liquid and the radius of the capillary (r). The equation relating these...
-
Chloramphenicol (at right) is a potent antibiotic, isolated from Streptomyces venezuelae, that is particularly effective against typhoid fever. It was the first naturally occurring substance shown to...
-
(a) Give the (R,S) designations for each chirality center in compound A and for compound B. (b) Write the Fischer projection formula for a compound C that is the diastereomer of A and B. (c) Would C...
-
(a) Is trans-1, 2-dimethylcyclopentane (5) superposable on its mirror image (i.e., on compound 6)? (b) Is cis-1, 2-dimethylcyclopentane (7) superposable on its mirror image? (c) Is cis-1,...
-
Determine your monthly payment if you buy a $300,000 house at 6% with 15% down and pay it off over 30 years.
-
Revenue Recognition at a Point in Time versus Revenue Recognition Over Time. The market company won a contract to build a shopping center at a price of $300 million. The following schedule details...
-
Calculate the flat monthly instalment for the car loan amount of $450,000 over two years at 5% compounded semiannualy. You can use either geometric progression or an optimizer (Goal seek or Solver)...

Study smarter with the SolutionInn App


