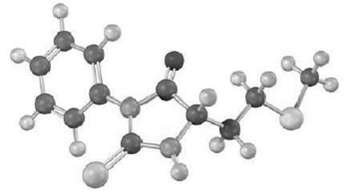
What is the N-terminal residue on a peptide that gives the following PTH derivative on Edmandegradation?
Question:
What is the N-terminal residue on a peptide that gives the following PTH derivative on Edmandegradation?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
The part of the PTH derivative that lies to the rig...View the full answer

Answered By

YOGENDRA NAILWAL
As I'm a Ph.D. student, so I'm more focussed on my chemistry laboratory. I have qualified two national level exams viz, GATE, and NET JRF (Rank 68). So I'm highly qualified in chemistry subject. Also, I have two years of teaching experience in this subject, which includes college teacher as well as a personal tutor. I can assure you if you hire me on this particular subject, you are never going to regret it.
Best Regards.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the n efficiency ? What is its range of possible values? Under what conditions will the efficiency be large?
-
The following data describe the catalysis of cleavage of peptide bonds in small peptides by the enzyme elastase. The arrow indicates the peptide bond cleaved in each case. a. If a mixture of these...
-
The result of Section 2.10, Exercise 25 gives the derivative of tan-1(x). Use integration by parts and a substitution to find tan-1(x)dx. Integration by parts along with substitution can be used to...
-
Once down to about 15, the worlds only wild flock of whooping cranes now numbers a record 237 birds in its Texas Coastal Bend wintering ground (www.SunHerald.com). The average whooping crane egg...
-
Can you express sympathy with someone's negative situation without apologizing for the circumstances? Explain your answer.
-
What alkyne would be useful for the synthesis of 2-heptanone (oil of cloves)?
-
In a reboiled absorber, operating as a deethanizer at 400 psia to separate a light hydrocarbon feed, conditions at the bottom tray are: Liquid Phase Molar flow = 1, \(366 \mathrm{lbmol} /...
-
The College Board reported the following mean scores for the three parts of the SAT (The World Almanac, 2009): Critical Reading ........502 Mathematics ..........515 Writing ............494 Assume...
-
Ten years ago Albert Phillips opened his own retail store and sold unpainted furniture. His store was located in Lakeside, a small city in the southeastern part of the United States. Although his...
-
Hooper Printing Inc. has bonds outstanding with 9 years left to maturity. The bonds have an 8% annual coupon rate and were issued 1 year ago at their par value of $1,000. However, due to changes in...
-
The octapeptide angiotensin II has the sequence Asp-Arg-Val-Tyr-IIe-His-Pro-Phe. What fragments would result if angiotensin II were cleaved with trypsin with chymotrypsin?
-
Draw the structure of the PTH derivative that would he formed on Edman degradation of angiotensin II (Problem 26.12).
-
On the first day of the fiscal year, Music Productions Corporation had 210,000 shares of $2 par common stock issued (at par) and outstanding, and the retained earnings balance was $900,000. Show how...
-
General Mills, the makers of Betty Crocker baking mixes and cereals such as Cheerios, had a contract with the local grocery store, FoodMart, where FoodMart would purchase boxes of cereal and various...
-
6. Find the displacement w(x, t) of an elastic semi-infinite string by solving the initial-boundary value problem aw(x,t) 2 w(x, t) = 2 " 0
-
Describe two (2) inclusion and diversity practices that have changed in recent years toward greater appreciation of inclusion and diversity in the workplace
-
Find the below questions for Finland. You may use the FRED, the World Development Indicators (WDI) database by the World Bank, OR the OECD. Stat database to respond to the following questions. 1)...
-
1. Paula purchased a frozen pizza at the grocery store and baked it in her oven. When she bit into the pizza, sudden pain shot through her mouth and she lost a large piece of her dental bridge, which...
-
Consider the following series of cash flows: Clearly set up an expression and determine the value of a lump sum equivalent amount of the above cash flows at year 5 if i = 7 percent. Use at least one...
-
A copper wire (density = 8.96 g/cm 3 ) has a diameter of 0.25 mm. If a sample of this copper wire has a mass of 22 g, how long is the wire?
-
The binding of oxygen by hemoglobin in the blood involves the equilibrium reaction: In this equation, Hb is hemoglobin. The pH of normal human blood is highly controlled within a range of 7.35 to...
-
Explain why the enol form of phenol is more stable than the keto form of phenol (eq. 9.43).
-
Draw the keto and enol tautomers of 1,3-diphenyl-1,3-propanedione.
-
Lily aldehyde, used in perfumes, can be made starting with a mixed aldol condensation between two different aldehydes. Provide their structures. (CH3)C CH,CHCH= O CH3 lily aldehyde
-
Perform one step of row reduction, in order to calculate the values for x and y by back substitution. Then calculate the values for x and y. Also calculate the determinant of the original matrix....
-
1 23 13 23 -3 23 23 23 23 Let A be the matrix of the quadratic form below. It can be shown that the eigenvalues of A are 6, 9, and 12. Find an orthogonal matrix P such that the change of variable x =...
-
Solve for X. -2] 9 2 x + 1 } = 3 3 x [5

Study smarter with the SolutionInn App


