What is wrong with these reactions explain. CI + NaOCH3 OCH3 + NaCl a) + HBr
Question:
What is wrong with these reactions explain.
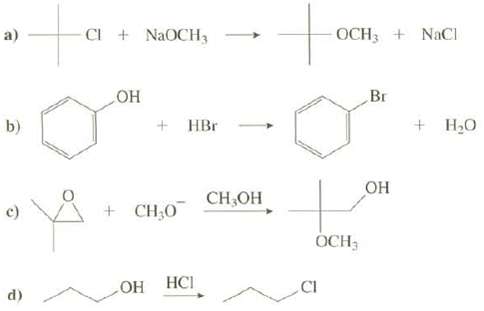
Transcribed Image Text:
CI + NaOCH3 OCH3 + NaCl a) но + HBr Br Н.о b) он CH,OH + CH;0 OCH3 НС CI ОН d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a The product shown will only be a very minor product because the reaction of a tertiary alkyl ...View the full answer

Answered By

Ankur Gupta
I have a degree in finance from a well-renowned university and I have been working in the financial industry for over 10 years now. I have a lot of experience in financial management, and I have been teaching financial management courses at the university level for the past 5 years. I am extremely passionate about helping students learn and understand financial management, and I firmly believe that I have the necessary skills and knowledge to effectively tutor students in this subject.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is wrong with these synthesesexplain. 1) NaNH2, NH, (1) C=CCH3 a) CH3C=CH 2) -Br CH;CH,-NH, I b) CH;CH,I + NH3 CI + Br OCH, CH, + CH,O d) . .. Br + HBr e) CH3 CH3 H,SO, f)
-
What is wrong with the caption of the cartoon shown in Figure Q36.1?
-
What is wrong with these names? Provide the correct name for each. (a) 5, 5 Dimethyl-3-ethylhexane (b) 2-Dimethyl pentane
-
Define pros and cons of the current transportation strategy of BEST and elaborate recommendations.
-
Indicate, for each of the costs listed below, whether it is: (a) Product cost (b) Period cost (c) Conversion cost (d) Prime cost (e) Direct material cost (f) Direct labour cost (g) Manufacturing...
-
According to the University Of Nevada Center for Logistics Management, 6% of all merchandise sold in the United States gets returned (BusinessWeek, January 15, 2007). A Houston department store...
-
Hulus Hybrids has the following information for the years ending January 31 2010 and 2009: Requirements 1. Compute the rate of inventory turnover for Hulus Hybrids for the years ended January 31,...
-
Selected transactions completed by ATV Discount Corporation during the current fiscal year are as follows: Jan. 5. Split the common stock 4 for 1 and reduced the par from $20 to $5 per share. After...
-
Garden Sales, Incorporated, sells garden supplies. Management is planning its cash needs for the second quarter. The company usually has to borrow money during this quarter to support peak sales of...
-
A study reports an analysis of 35 key product categories. At the time of the study, 72.9% of the products sold were of a national brand, 23% were private label, and 4.1% were generic. Suppose that...
-
Show how this synthesis might beaccomplished: Br CH3 CH3 HO from "CN -
-
Show all the steps in the mechanisms for these reactions. Don't forgot to use curved arrows to show the movement of electrons in eachstep. CH3 CH3 ) . + CHH ., + Br CH; CH3 H,SO, CH;CH,OCH,CH; + H;O...
-
A mail-order prescription drug vendor measured the number of errors per standard order being picked in their distribution center. Data can be found in the worksheet C16P11 in the OM6 Data Workbook ....
-
2. Free-Evaporation measurements The standard US Class A evaporation pan has an inside diameter of 47.5 inches and a depth of 10 inches. a. Calculate the surface area of water in the pan in square...
-
Q9-) The following measurements are from an experiment. F (N) is force which is acting on an object, and a (m/s) is the acceleration of this object. Answer the questions about given graphics. a)...
-
Royal Lawncare Company produces and sells two packaged products-Weedban and Greengrow. Revenue and cost Information relating to the products follow: Selling price per unit Variable expenses per unit...
-
To shake her attacker, Woody flies directly upwards at 2 1 . 0 km / h ( woodpeckers can fly slightly faster than this when flapping their wings, but this particular woodpecker is a little weighted...
-
Identify two nontheatrical events that require people to engage in imitation or role - playing. How is imitation used in these situations? What makes these situations different from theater?
-
When a machine tool is placed directly on a rigid floor, it provides an excitation of the form \[ F(t)=(4000 \sin 100 t+5100 \sin 150 t) \mathrm{N} \] to the floor. Determine the natural frequency of...
-
The domain of the variable in the expression x 3/x + 4 is________.
-
Simplify each expression. --7-p-3)-3(3p+8)
-
The reaction of 1-chlorooctane with CH3CO2 to give octyl acetate is greatly accelerated by adding a small quantity of iodide ion. Explain.
-
Compound X is optically inactive and has the formula C16H16Br2. On treatment with strong base, X gives hydrocarbon Y, C16H14. Compound Y absorbs 2 equivalents of hydrogen when reduced over a...
-
When a primary alcohol is treated with p-toluenesulfonyl chloride at room temperature in the presence of an organic base such as pyridine, a tosylate is formed. When the same reaction is carried out...
-
There are n undergraduate students and k departments at some university. The Student Senate must have k students, one from each department. It also should have k freshmen, k sophomores, kg juniors,...
-
what are Total Liabilities equate to 478,523,226? Debt to Capitalization Ratio = (Total Liabilities (Total Assets + Total Equity)) x 100 Debt to Capitalization Ratio =...
-
Victor Mineli, the new controller of Wildhorse Co., has reviewed the expected useful lives and salvage values of selected depreciable assets at the beginning of 2025. Here are his findings: Type of...

Study smarter with the SolutionInn App


