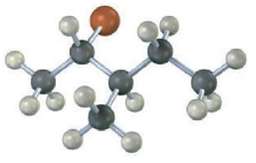
Question: What stereo chemistry do you expect for the tri-substituted alkene obtained by E2 elimination of the following alkyl halide on treatment with KOH? (Reddish brown
What stereo chemistry do you expect for the tri-substituted alkene obtained by E2 elimination of the following alkyl halide on treatment with KOH? (Reddish brown =Br.)
Step by Step Solution
★★★★★
3.46 Rating (178 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

Strategy As in the previous problem draw the structure convert it to a Newma... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
22-C-O-E-R (111).docx
120 KBs Word File


