Question: When a carboxylic acid is dissolved in isotopically labeled water, the label rapidly becomes incorporated into both oxygen atoms of the carboxylic acid. Explain. R.
When a carboxylic acid is dissolved in isotopically labeled water, the label rapidly becomes incorporated into both oxygen atoms of the carboxylic acid. Explain.
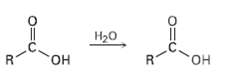
R. , R.
Step by Step Solution
★★★★★
3.39 Rating (158 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

R OH HO HO O OH OH R OH HO T The tetrahedral intermedi... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
22-C-O-CA (144).docx
120 KBs Word File


