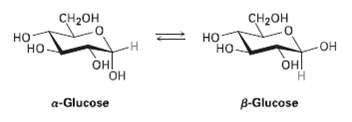
When crystals of pure ?-glucose are dissolved in water, isomerization slowly occurs to produce ?-glucose. Propose a
Question:
When crystals of pure ?-glucose are dissolved in water, isomerization slowly occurs to produce ?-glucose. Propose a mechanism for the isomerization.
Transcribed Image Text:
CH2он CH2он но- -Н но но но ОН он он Он Н B-Glucose a-Glucose
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)
Some hydrogens have been omitted to make the drawing less cluttered CHOH CHOH 0 HOH ...View the full answer

Answered By

Daniel Kiunga
I am a highly motivated individual with a track record of scholastic achievement. I hold a Master of Science from a reputable institution. I can handle the following subjects:
Business and Management
Law
Health sciences and Nursing
Biology
History
Marketing
Social and political sciences.
I have over 8 years experience in academic writing. I am familiar with APA, MLA, CHICAGO, and HAVARD styles. My aim is to use my research skills to help students achieve the best by providing top-class academic scripts.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the triolefin process using a metal alkylidene as the catalyst. CH IM CHCH CH - - - CH3 propylene but-2-ene (cis + trans) ethylene
-
Propose a mechanism for the following reaction: CI Alcl
-
Propose a mechanism for the following reaction. (Number the carbons to help you see where they end up in the product.) CH3 CH2 H,c ,504 CH3 CH2 CHCH2CH2CH CCH3 CH3COH
-
In Figure P23.15, determine the point (other than infinity) at which the electric field is zero. 1.00 m -2.50 : 6.00 C
-
1. What should Angela have done to prepare herself for telework? 2. What should Ralph have done to prevent this problem from occurring?
-
Ellen wants to know how many books each author wrote. Write a SQL statement to provide such information to Ellen? Use the following information: Ellen Novotny started an online bookstore in 2017. You...
-
The table below describes a hypothetical experiment on 2400 persons. Each row of the table specifies a category of person, as defined by their pre-treatment predictor $x$, treatment indicator $T$,...
-
After the tangible assets have been adjusted to current market prices, the capital accounts of Brandon Newman and Latrell Osbourne have balances of $75,000 and $125,000, respectively. Juan Rivas is...
-
Consider a 1.0 m guitar string. At what distances from the end should you pluck the string if you want to enhance the n = 4 mode? Is n = 4 the only mode that is excited? Explain.
-
Each of the following cost items pertains to one of the following companies: Toyota (a manufacturing-sector company), Sobeys (a merchandising-sector company), and Google (a service-sector company):...
-
Primary amines react with esters to yield amides: RCO2R' + R''NH2 ? RCONHR'' + R'OH. Propose a mechanism for the following reaction of an ?, ? un-saturated ester. CH .CH + CH3NH2 H3C- + CH3OH CO2CH3
-
When glucose (Problem) is treated with NaBH4, reaction occurs to yield sorbitol, a polyalcohol commonly used as a food additive. Show how this reductionoccurs. CH- NABHA -2 Sorbitol Glucose
-
Provide an example of each of the three types of models that might apply to designing a car, a house, and an office building. Explain why requirements models are logical models rather than physical...
-
A recent accounting statement showed the following: Raw material (RM): Work in process (WIP): Finished goods (FG): Total sales revenue (R): 529,073 2,999,396 3,543,222 71,132,934 Cost of goods % of...
-
Considering the principles of abstraction and encapsulation, discuss the advantages and disadvantages of implementing functions as first-class citizens within programming languages, and how this...
-
Describe the composition of the courtroom work group. Explain how it controls the processing of criminal cases in most jurisdictions. Please provide as much details as possible.?
-
Janina, Incorporated, has the following mutually exclusive projects. Year Project A Project B 0 $ 3 3 , 0 0 0 $ 3 6 , 0 0 0 1 1 8 , 5 0 0 1 9 , 5 0 0 2 1 5 , 0 0 0 1 3 , 5 0 0...
-
What role does memoization play in optimizing computational performance within recursive functions, and how does it enhance efficiency by storing previously computed results for future reference?
-
Consider a functional of the form \[T(F)=\int_{-\infty}^{\infty} \int_{-\infty}^{\infty} h\left(x_{1}, x_{2}ight) d F\left(x_{1}ight) d F\left(X_{2}ight)\] where \(F \in \mathcal{F}\), a collection...
-
Let (X. A. p) be a measure space. Show that for any A,B A, we have the equality: (AUB)+(An B) = (A) + (B).
-
Consider the phase diagram for iodine shown here. a. What is the normal boiling point for iodine? b. What is the melting point for iodine at 1 atm? c. What state is present at room temperature and...
-
When 2-bromo-1, 3-dimethylbenzene is treated with sodium amide in liquid ammonia, no substitution takes place. This result can be interpreted as providing evidence for the elimination-addition...
-
(a) Outline a step-by-step mechanism for the phenylation of acetoacetic ester by bromobenzene and two molar equivalents of sodium amide. (Why are two molar equivalents of NaNH2 necessary?) (b) What...
-
Rank the following in order of increasing acidity. NO2 CF3 CF3
-
Pangasa Co. paid its annual worker accident insurance premium of $48,000 on its manufacturing facility in January. The company expects two worker accidents to happen during the year, and to make...
-
3. The game of baseball is often analyzed using Markov models. The state of the game can be represented by listing bases that have runners and keeping track of how many outs there are. There are...
-
Thornton Electronics currently produces the shipping containers it uses to deliver the electronics products it sells. The monthly cost of producing 9,300 containers follows. Unit-level materials...

Study smarter with the SolutionInn App


