Question: Primary amines react with esters to yield amides: RCO2R' + R''NH2 ? RCONHR'' + R'OH. Propose a mechanism for the following reaction of an ?,
Primary amines react with esters to yield amides: RCO2R' + R''NH2 ? RCONHR'' + R'OH. Propose a mechanism for the following reaction of an ?, ? un-saturated ester.
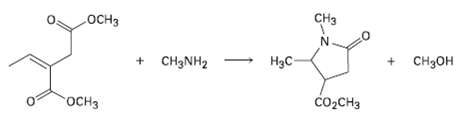
CH .CH + CH3NH2 H3C- + CH3OH CO2CH3
Step by Step Solution
3.39 Rating (165 Votes )
There are 3 Steps involved in it
CH3NH H3C CHO CH3O CHOH HC HC loss of methanol CH3NH HC conjugate addition of methy... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-AN (117).docx
120 KBs Word File


