Which, if any, of the following compounds can be prepared by an acetoacetic ester synthesis?Explain. (a) Br.
Question:
Which, if any, of the following compounds can be prepared by an acetoacetic ester synthesis?Explain.
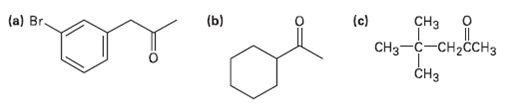
Transcribed Image Text:
(a) Br. (c) (b) CHз CHз —с—сн-ссНз CHз чнорио
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Look back to Problem 2214 which describes compounds that can be prepare...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following compounds can be prepared by radical halogenation with little complication by formation of isomeric by-products? CI CI
-
Which, if any, of the following compounds can be prepared by a malonic ester synthesis? Show the alkyl halide you would use in each case. (a) Ethyl pentanoate (b) Ethyl 3-rnethylbutanoate (c) Ethyl...
-
Show how each of the following compounds can be prepared from cyclohexene: a. b. c. d. OH
-
CMOS Chips is hedging a 20-year, $21 million, 8% bond payable with a 20-year interest rate swap and has designated the swap as a fair value hedge. The agreement called for CMOS to receive payment...
-
Recline in your first-class seat and sip a freshly stirred drink while listening to 12 channels of superb audio, or snooze is an example of what type of persuasive appeal? How does it compare to the...
-
Describe situations where each of the different isolation levels would be useful for transaction processing.
-
Six test scores are shown below. The first 4 test scores are 15% of the final grade, and the last two test scores are 20% of the final grade. Find the weighted mean of the test scores. 80 70 84 93 89...
-
Johnson, Larson, and Kragen own an advertising agency that they operate as a partnership. The partnership agreement includes the following: a. Johnson receives a salary of $50,000. b. Larson receives...
-
If a sensor (I1:0) is used to count the incoming cars, and another sensor (I1:1) is used to count the outgoing cars, please design a PLC program such that when the number of cars in the parking lot...
-
Schank Marketing Research has just signed contracts to conduct studies for four clients. At present, three project managers are free for assignment to the tasks. Although all are capable of handling...
-
Predict the product(s) of the followingreactions: (b) 1. Nat "OEt Co (a) 2. CH3I eat (c) Br2, PBr3 H20 CH-CH2C (d) " NaOH, H20 12
-
How would you prepare the following ketones using an acetoacetic estersynthesis? (a) (b) CH-H CH-CH CH2CH2CHCH3 CH3 CH
-
List the following substances in order of increasing molar entropy at 298 K and 1 bar: NH 3 (g), HF(g), H 2 O(s), NH 2 OH(g). Explain your reasoning.
-
What is meant by internal consistency when applied to forecasting assumptions? Give an example of internal inconsistency.
-
On September 1, Global Tech Inc. purchased merchandise for resale for $20,800 on credit terms 2/15, n/60 using the gross method and a perpetual inventory system. Global Tech incurred a shipping...
-
Identify the reasons that a health care organization needs cash on hand.
-
In what ways might the issue addressed in the research paper affect you as you start your career? What are the potential consequences of reliance on specialists to assist the auditor in making high...
-
Explain the functionality of the C++ code. Use the C++ to Assembly Activity Template to complete thisstep. The C++ file is located within the Software Reverse EngineeringPlayground in the Module One...
-
Construct a frequency and relative frequency histogram of the five-year rate of- return data discussed in Example 3. Approach To draw the frequency histogram, use the frequency distribution in Table...
-
On October 31 Juanita Ortega, owner of Outback Guide Service, received a bank statement dated October 30. Juanita found the following: 1. The checkbook has a balance of $2,551.34. 2. The bank...
-
What is a chemical bond according to valence bond theory?
-
Give the products (if any) when each of the following isomers reacts with HBr and heat. CH OH OCH 3-(hydroxymethyl)phenol 3-methoxyphenol
-
What products (if any) are formed when 3, 5-dimethylbenzenethiol is treated first with one equivalent of Na+ C2H5O- in ethanol, and then with each of the following? (a) Allyl bromide (b) Bromobenzene
-
What products (if any) are formed when 3, 5-dimethylbenzenethiol is treated first with one equivalent of Na+ C2H5O- in ethanol, and then with each of the following? (a) Allyl bromide (b) Bromobenzene
-
KORBIN COMPANY Comparative Income Statements For Years Ended December 31 2021 2020 $ 316,588 2019 $ 219,700 Sales Cost of goods sold Gross profit Selling expenses Administrative expenses Total...
-
Following are the capital account balances and profit and loss percentages (indicated parenthetically) for the Alden, Boyd, and Carmel partnership: Alden (40%) Boyd (40%) Carmel (20%) $ 220,000...
-
Required information E3-13 (Algo) Recording and Posting Accrual Basis Journal Entries, and Preparing an Unadjusted Trial Balance and Preliminary Financial Statements [LO 3-2, 3-3, LO 3-4] [The...

Study smarter with the SolutionInn App


