Question: Which of the following compounds have the same oxidation level, and which have differentlevels? OH 5.
Which of the following compounds have the same oxidation level, and which have differentlevels?
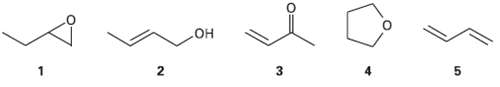
OH 5.
Step by Step Solution
3.45 Rating (171 Votes )
There are 3 Steps involved in it
OH Co 6 6 All ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-O-H (32).docx
120 KBs Word File


