Tell whether each of the following reactions is an oxidation, a reduction, orneither: (a) CH3CH CH3CH2OH
Question:
Tell whether each of the following reactions is an oxidation, a reduction, orneither:
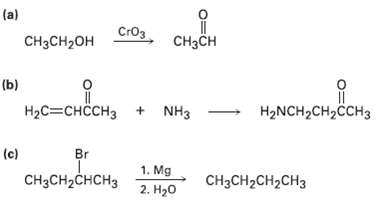
Transcribed Image Text:
(a) Стоз CH3CH CH3CH2OH (b) HаС—снссHз + NHз — НаNCH2CH2CCH3 Br (c) 1. Mg CHзCH2CHCHз CняCH2CH2CH3 2. H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a This reaction is an oxidation b The reac...View the full answer

Answered By

Arshad Ahmad
Well, I am really new to tutoring but I truly believe a good student can be a better teacher. I have always been a topper at school. I passed my Chartered Accountancy at a very young age of 23, a rare feat for most of the students. I am really dedicated to whatever work I do and I am very strict regarding deadlines. i am always committed and dedicated to whatever work allotted to me and I make sure it is completed well within deadline and also I try to give my best in whatever I do. Hope we will have a good time studying together.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Indicate whether each of the following reactions is an oxidation reaction, a reduction reaction, or neither: a. b. c. d. e. f. H2 CHj 3CI partially deactivatedCH Pd HBr RCH CHRRCH2CHR Br2 H2CrO4...
-
Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB, orE2: NaN3 (a) CHCH2CH2H2Br CH3CH2CH2CH,N=N=N THE CI (b) , CCH-CHCH2CH CHCH2CHCHCH3 Ethanol (c) H CI -CH -- -CH (d)...
-
Tell wbether each of the following reactions favors reactants or products at equilibrium. (Assume that all reactants and products are soluble.) (a) CH3CI + I- CH3I + CI- (b) CH3CI + -OCH3 CH3OCH3 +...
-
Scranton Refrigeration Corporation began operations at the beginning of the current year. One of the companys products, a compressor, sells for $370 per unit. Information related to the current years...
-
Refer to the BMW Company case. Design a spreadsheet that will allow the firm's managers to estimate what percentage of the firm's net income 10 years into the future will be devoted to disposal of...
-
Use superposition to find vo in the circuit of Fig. 4.81. Figure 4.81 For Prob.4.13 4 A 8 12 V 2 A 10
-
Cloud Tech Ltd produces four types of drones, which are sold to local retail stores. The marketing department expects the following sales performance for the next year. Required Prepare a monthly...
-
Long Corporation is a fabric manufacturing company. On January 20, Long Corporation made sales to Lyndsays Lace in the amount of $15,000 with terms of 2/10, n/30. Lyndsays Lace paid Long Corporation...
-
Examine advanced synchronization mechanisms in RTOS, such as priority inheritance and real-time mutexes. How do these mechanisms contribute to maintaining real-time performance ?
-
A liquid mixture of benzene and toluene is to be separated in a continuous single-stage equilibrium flash tank. The pressure in the unit may be adjusted to any desired value, and the heat input may...
-
Which of the following compounds have the same oxidation level, and which have differentlevels? OH 5.
-
How would you carry out the followingsyntheses? Cyclohexene Cyclohexanol Cyclohexane ~/~/al
-
Suppose that the UDP receiver computes the Internet checksum for the received UDP segment and finds that it matches the value carried in the checksum field. Can the receiver be absolutely certain...
-
The financial information in the Excel file associated with this exercise was obtained from Form 10-K reports for Costco Wholesale Corporation . In this exercise, we examine how changing the starting...
-
Complete all questions 1. Refer to the "Caterpillar" file in Moodle and answer the following. a. Calculate the Addition Funds Needed for Working Capital for Caterpillar for the calendar year 2016. b....
-
1. Prepare journal entries to record the following transactions for Sherman Systems. a. Purchased 6,900 shares of its own common stock at $44 per share on October 11. b. Sold 1,475 treasury shares on...
-
Pick any two leadership styles that interests you out of the following leadership styles: Autocratic Leadership Bureaucratic Leadership Democratic Leadership Laissez-faire Leadership ...
-
Evaluating if most suitable leadership approaches have been used and if they were used correctly and sufficiently for seven eleven 7/11. Instruction: please provide thorough explanation that is...
-
How might HBOs organizational culture change as a result of the changes described in this case?
-
Flicker, Inc., a closely held corporation, acquired a passive activity this year. Gross income from operations of the activity was $160,000. Operating expenses, not including depreciation, were...
-
The heat of atomization is the heat required to convert a molecule in the gas phase into its constituent atoms in the gas phase. The heat of atomization is used to calculate average bond energies....
-
Draw Lewis structures for the following free radicals. (a) The ethyl radical, (b) The tert-butyl radical, (CH3)3C (c) The isopropyl radical (2-propyl radical) (d) The iodine atom CH3 CH2
-
(a) Using bond-dissociation enthalpies from Table 4-2 (page 143), calculate the heat of reaction for each step in the free-radical bromination of methane. (b) Calculate the overall heat of reaction....
-
The reaction of tert-butyl chloride with methanol Is found to follow the rate equation Rate = kf [(CH3)3C-Cl] (a) What is the kinetic order with respect to tert-butyl chloride? (b) What is the...
-
A 200 mm reinforced concrete wall carries a dead load of 140 kN/m including self-weight and a live load of 60 kN/m. It is to be carried by 500 mm thick footing with its bottom 1.5 m below NGL. Soil...
-
Using www.land.vic.gov.au look up your current place of abode to gather planning information that might be useful in assessing its future development potential. 2. Write one-page report in WORD...
-
The following situation is an example for a fire detection and suppression system in an office building. The Initiating Event (IE) for the Event Tree is "fire starts", with a frequency of once every...

Study smarter with the SolutionInn App


