Which of these compounds would have a faster rate of SN2 reaction? a) b) c) d) e)
Question:
Which of these compounds would have a faster rate of SN2 reaction?
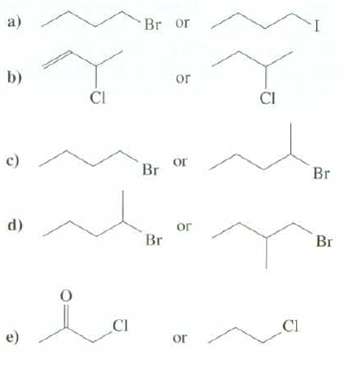
Transcribed Image Text:
a) b) c) d) e) CI CI Br or or Br or Br or or CI CI Br Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
a The right compound has a faster rate because iodide ion is a better leaving group than bromid...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain which compound has a faster rate of SN2 reaction: a) HC CI CI c) PhCHCH3 or H CI Cl or CHCHCH3 b) CI CH3 CH3 or U
-
Which of these compounds would have faster rate of SN1 reaction? a) b) c) d) e) CI ta Ph Ph -CI or Br or Br or or -Cl or + CH3 Ph -Cl Br Br ta -CI
-
Which of these compounds would you expect to have the highest boiling point? Explain. [Section 24.4] CH3CH CH CH OH CHC=CH HCOCH
-
A long cylindrical wood log (k = 0.17 W/mK and = 1.28 10 -7 m 2 /s) is 10 cm in diameter and is initially at a uniform temperature of 15C. It is exposed to hot gases at 550C in a fireplace with a...
-
Steelworx Ltd accumulates costs for its single product using weighted average process costing. Direct material is added at the beginning of the production process, and conversion occurs uniformly...
-
Projections indicated that there would be record numbers of homeless people over the next few years, so the city decided that it was important to make shelters available. Accordingly, contracts were...
-
A senior VP has proposed that "we pay too much tax, so we should issue new debt and use the proceeds to buy back common stock." What would be the logic behind this statement?
-
Water is to be pumped from a lake to a ranger station on the side of a mountain (see figure). The flow rate is to be 95gal/mm, and the flow channel is a standard 1-in. Schedule 40 steel pipe (ID =...
-
Research System Incident and Event Manager (SIEM). I need a basic description including what it is, how it helps security, and what devices should it monitor. Explain.
-
Mr B aged 52 years, has earned rupees 75,00,000 out of his business. His ex-wife gifted him a car worth rupees 8 lakh. He spent a total of rupees 20 lakh during a family trip. He won a lottery of 16...
-
(a) Show all of the steps in the mechanism for this reaction. Don't forget to use curved arrows to show the movement of electrons in each step of the mechanism. (b) Show a free energy versus reaction...
-
Arrange these compounds in order of increasing SN2 reaction rate: CI Br CI Br
-
Assume that a researcher had 14 individuals participate in all three conditions of her experiment. Use this information to complete the source table below. df MS F Source SS Between 60 Subjects...
-
Light a matchstick by striking at the edge of the match box. If the event takes place in a room that can be considered a closed system, what are the consequences? If the same event occurs inside an...
-
In javelin throw, an athlete usually runs a certain distance before the throw. Why does the athlete not throw their javelin from a stand-still position?
-
A Labrador retriever dog with inertia \(25 \mathrm{~kg}\) and velocity \(6 \mathrm{~m} / \mathrm{s}\) jumps into a trolley at rest and after that both move in the same direction with a velocity \(3...
-
Do you think this is an issue worth generating conflict over? What are the potential costs and benefits of Matt and Peter having an open discussion of the issues?
-
As a team member, do you believe you have a responsibility to act as a mediator to ensure the conflict does not impact the teams success?
-
Example 2.14 introduced the design equation for a countercurrent hollowfiber boiler feed water deaerator when all the resistance to mass transfer resides on the liquid phase and the entering gas...
-
On January 1, 2018, Khalid Ltd., which follows IAS 17, entered into an eight-year lease agreement for three dryers. Annual lease payments for the equipment are $28,500 at the beginning of each lease...
-
Draw a box-and-whisker plot for each data set. 4 3 Hits in a Round of Hacky Sack 12 3 3 5 18 1 19 6 3
-
Rank each of the following series of compounds in order of increasing oxidationlevel: CI (a) (b) CH3CN CH3CH2NH2 H2NCH2CH2NH2
-
Tell whether each of the following reactions is an oxidation, a reduction, orneither. (a) NABH4 H20 CH;CH- CH3CH2CH2OH (b) OH 1. BH3 2. NaOH, H202
-
Give a JUPAC name for each of the following alkyl halides (yellow green =Cl): (b) (a)
-
As a precaution, the levers on gasoline pumps in many provinces in Canada must be manually operated at all times. One of the reasons for this is especially relevant in the winter time because the...
-
7. A transformer core is shown in cartoon form in Figure 8.26. This is made up of, perhaps implausibly, 29 gage M-36 laminations: the same sort of material described by Figures 8.19 and 8.20. Assume...
-
2. For a lamp that has a total luminous flux, of 4.5104 lm and total radiant flux, of 300 W, (a) what is the overall luminous efficiency, K for this lamp? (b) if this lamp has a luminous intensity of...

Study smarter with the SolutionInn App


