Give a JUPAC name for each of the following alkyl halides (yellow green =Cl): (b) (a)
Question:
Give a JUPAC name for each of the following alkyl halides (yellow green =Cl):
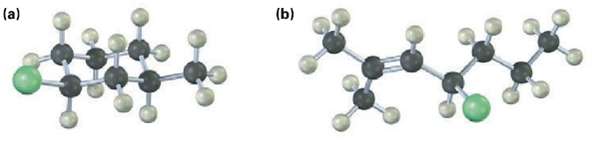
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a CH3 C CH3 CH3...View the full answer

Answered By

ALBANUS MUTUKU
If you are looking for exceptional academic and non-academic work feel free to consider my expertise and you will not regret. I have enough experience working in the freelancing industry hence the unmistakable quality service delivery
4.70+
178+ Reviews
335+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give the IUPAC name for each of the following alkyl groups, and classify each one as primary, secondary, or tertiary: (a) CH3(CH2)10CH2-- (b) (c) --C(CH2CH3)3 (d) (e) (f) -CH2CH2CHCH2CH2CH3 CH2CH3...
-
Give the IUPAC name for each of the following alkyl groups, and classify each one as primary, secondary, or tertiary: (a) CH3 (CH2)10CH2 (b) CH2CH2CHCH2CH2CH3 W CH2CH3
-
Give a substitutive name for each of the following compounds. (a) CH3CHT-O-CH'CH'-OH (b)
-
The MRP gross requirements for Item A are shown here for the next 10 weeks. Lead time for A is three weeks and setup cost is $ 10. There is a carrying cost of $ 0.01 per unit per week. Beginning...
-
Consider the stylized example of Figure 7.3, in which there is a jump in the mean value. Using the same worksheet as a template, enter the exponential smoothing calculations for these periods. Assume...
-
Find the mesh currents in the circuit of Fig. 3.98 using MATLAB. Figure 3.98 For Prob. 3.53 Is WA I3 3 mA 1 k2 4 k2 12 V
-
All Aboard Ltd is preparing a quarterly budget covering the 3 months ending 30 June 2025. The information available for the budget is as follows. 1. Cash sales represent 40% of all monthly sales; 60%...
-
During 2019, Jason and Vicki Hurting, who are married with two children and filing jointly, had the following tax information. Jason owns a cash-basis landscaping business, and Vicki works as a sales...
-
Jim Carrie Company shows a balance of $221,140 in the Accounts Receivable account on December 31, 2025. The balance consists of the following. Installment accounts due in 2026 $23,000 Installment...
-
Figure 14P-1 presents the demand curve, marginal revenue, and marginal costs facing a monopolist producer. a. What is the profit-maximizing level of output? b. What price will the monopolist charge...
-
Tell whether each of the following reactions is an oxidation, a reduction, orneither. (a) NABH4 H20 CH;CH- CH3CH2CH2OH (b) OH 1. BH3 2. NaOH, H202
-
Show the product(s) of reaction of the following alkenes withNBS: (b) (a)
-
What is the discriminant and what information does it provide about a quadratic equation?
-
Requirement : How to calculate the Theoretical hp of Venturi Meter. Velocity can be calculated as below: By V4=Qt/a4 Run 1 2 3 4 45 5 6 7 8 9 10 Tube No. 1 2 3 4 5 ( 1 8 9 10 11 Qt (m/s) 0.4811312386...
-
How are you doing with practicing using the art of silence effectively as an elementary school counselor intern ?
-
Charles and Company has begun selling a new pie recipe and they want you to help them with next year's budgeted financial statements.Using the worksheet below, complete Charles and Company forecast...
-
Jill purchased her principal residence on January 1, Year 5, for $280,000. On January 1, Year 6, when it was worth $250,000, she turned it into a rental house. On January 1, Year 9, after...
-
What are examples of the three leadership styles: Servant Leadership, Visionary Leadership, and Empathetic Leadership?
-
Find the probability of randomly selecting 2 different challenges and finding that they were both accepted challenges made by female players. Use the following results from the 839 player challenges...
-
Maria Castigliani is head of the purchasing department of Ambrosiana Merceti, a medium-sized construction company. One morning she walked into the office and said, The main problem in this office is...
-
The titration of 10.00 mL of HCl solution of unknown concentration requires 12.54 mL of a 0.100 M NaOH solution to reach the equivalence point. What is the concentration of the unknown HCl solution...
-
Construct a graph, similar to Figure 3-11, of the torsional energy of 3-methylpentane along the C2-C3 bond. Place C2 in front, represented by three bonds coming together in a Y shape, and C3 in back,...
-
The following names are all incorrect or incomplete, but they represent real structures. Draw each structure and name it correctly. (a) 2-ethylpentane (b) 3-isopropylhexane (c)...
-
Provide IUPAC names for the following compounds. (a) (CH3)2CHCH2CH3 (b) CH3-C(CH3)2-CH3 (c) (d) (e) (f) CH CH CHCH CH,CHCHCH le ' CH CH,CH, CH CH CH, CH CH C(CH CH,CH,CHCHCH, CH(CH2 CH CHCH,CH, CH)C...
-
A car is traveling around a banked curve with a radius of 100 meters and a banking angle of 30 degrees at a speed of 40 m/s. What is the coefficient of friction between the tires and the road that...
-
A spring has a force function of F(x) = 3x, where x is the displacement from the equilibrium position. If an object with a mass of 2 kg is moved from x = 0 to x = 4 meters, how much work is done by...
-
A crane lifts a load with a force of 1000 N over a distance of 10 meters in 20 seconds. What is the average power generated by the crane?

Study smarter with the SolutionInn App


