Question: Which of these groups has the higher priority? b) -COH or CNH2 a) -CH3 or -CH,CH3 c) -CH;CH,CH3 d) -CH-CH CH3 or -C=N or -CH;CH,CH,CH3
Which of these groups has the higher priority?
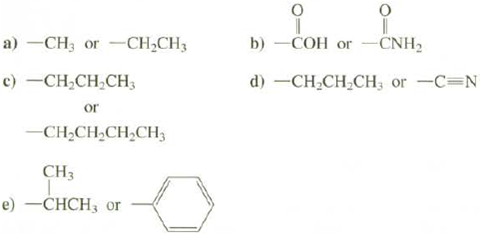
b) -COH or CNH2 a) -CH3 or -CH,CH3 c) -CH;CH,CH3 d) -CH-CH CH3 or -C=N or -CH;CH,CH,CH3 CH3 e) -CHCH3 or
Step by Step Solution
3.42 Rating (155 Votes )
There are 3 Steps involved in it
The CahnlngoldPrelog sequence rules are used to assign priorities to groups These rules use the atom... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-S (48).docx
120 KBs Word File


