Will either of the following reactions take place as written, according to the data inTable? (a) HCN
Question:
Will either of the following reactions take place as written, according to the data inTable?
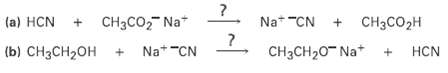
Transcribed Image Text:
(a) HCN + CH3CO2 Na+ Na* "CN CHзCO2H Na* "CN CH3CH20- Na* |(b) CH3CH2OH HCN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Strategy Use Table to find the strength of each acid A reaction take...View the full answer

Answered By

PRINCE PANDEY
I am Indian Chartered Accounting having a strong hold in the subjects of Accounting, IFRS Reporting, Indian
Taxation, Cost Accounting, Auditing. I have vast experience of teaching a student with easy way problem-solving approach.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following reactions take place in a batch reactor: A + B ( C (desired product) B + C ( D (hazardous product) As the reaction proceeds, D builds up in the reactor and could cause an explosion if...
-
The enthalpy change for each of the following reactions was calculated using bond energies. The bond energies of XO, YO, and ZO are all equal. XX + O=O XOOX; H = 275 kJ YY + O=O YOOY; H = +275 kJ...
-
As discussed in Section 6.2, either of the following two equations can be applied to determine the amount necessary to recover invested capital and a required return: CR 1 = P(AP,i,n) + S(AF,i,n) or...
-
Differentiate between formal and behavioral roles, and describe how behavioral roles emerge during group interaction.
-
Melissa owns a residential lot in Spring Creek, Louisiana, that has appreciated substantially in value. She holds the lot for investment. She is considering exchanging the lot for a residential lot...
-
Which business cost will increase as a restaurants volume increases? a. Napkin costs b. Mortgage cost c. Lighting costs d. Insurance costs
-
Use the forward equation to find the spot rate curve for the lattice constructed in Exercise 1. Data from Exercises 1 Construct a short rate lattice for periods (years) 0 through 9 with an initial...
-
The city of Morristown operates a printing shop through an Internal Service Fund to provide printing services for all departments. The Central Printing Fund was established by a contribution of...
-
At the beginning of 20x7, the accounts receivable had a beginning balance of $20,000 and allowance for doubtful accounts had a beginning credit balance of 3,000. The following information occurs...
-
Investigate the case of Makayla Sault, the 11 year-old Ontario First Nation girl who, with her parents, refused treatment for her acute lymphoblastic leukemia (ALL), and who subsequently died. Do you...
-
Amide ion, H 2 N , is a stronger base then hydroxide ion, HO . Which is the stronger acid, NH 3 or H 2 O? Explain.
-
Ammonia, NH3, has pKa = 36 and acetone has pKa 19. Will the following reaction takeplace? Na* -:NH2 NH3 CH H Acetone Na+ CH2:
-
Financial accountants use estimates of financial value differently than either cost or management accountants. REQUIRED 1. Identify two differences in use. 2. Identify a similarity among accountants.
-
How do bacterial quorum sensing mechanisms regulate population-wide behaviors and virulence factors, and what implications does this hold for therapeutic interventions targeting microbial communities?
-
An Operating System (OS) is an interface between a computer user and computer hardware. An OS performs all the basic tasks, like file management, memory management, process management, and the...
-
Compare and contrast cross-cultural preparation programs and diversity training programs. What would you include as the content of each program? Be specific and provide examples to illustrate your...
-
Which protein assay is most sensitive?
-
A city bus collected $780 infares on one day. The price of a regular fare was $0.80, and the price of a discount fare was $0.40. If a total of 1200 people paid the fares on this bus, how many people...
-
Can these views be logically reconciled?
-
Match each of the key terms with the definition that best fits it. _______________ A record of the sequence of data entries and the date of those entries. Here are the key terms from the chapter. The...
-
a. Calculate the fugacity of liquid hydrogen sulfide in contact with its saturated vapor at 25.5C and 20 bar. The vapor pressure of pure water at 310.6 K is 6.455 kPa. Compute the fugacity of pure...
-
Name these compounds: CH3 CH3 Br a) CHCHCHC-CHCHCH3 CH3 b)
-
Draw the structure of 3-bromo-4-butylcyclohexene.
-
Name these compounds: a) CHCHCHCH3 c) -CHCHCH b) CHCHCCHCHCH CH d) Br CH3
-
What advantage does the private resident buying oce (store-owned offices) aord its stores that independent buying oces do not? Describe two functions of an independent buying office. Describe the 4...
-
10. Consider the following data - == - -5 1 3 4 -1 0 1 1 (a) Find the Least Squares Regression Line. (b) Find the coefficient of correlation and determination. (c) Find a 90% confidence interval for...
-
Which point on the graph y=4x are closest to the point (0, 2)? (Hint: consider the distance formula between two points)

Study smarter with the SolutionInn App


