Ammonia, NH3, has pKa = 36 and acetone has pKa 19. Will the following reaction takeplace? Na*
Question:
Ammonia, NH3, has pKa = 36 and acetone has pKa 19. Will the following reaction takeplace?
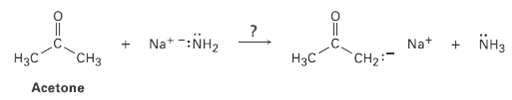
Transcribed Image Text:
Na* -:NH2 NH3 CHз Нас Hзс Acetone Na+ CH2:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
CH3 NaNH HC pK 19 Stro...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Under standard conditions, will the following reaction proceed spontaneously as written? Fumarate NADHHsuccinate NAD 1
-
Under standard conditions, will the following reaction proceed spontaneously as written? Cyto a(Fe + cyto b(Fe) oto a(Fe")+ ono b(Fe) 3 +
-
Ammonia and hydrogen chloride react to form solid ammonium chloride: NH3 (g) + HCl (g) NH4CL (s) Two 2.00-L flasks at 25oC are connected by a valve, as shown in the drawing on the next page. One...
-
Find a dense document that has little white space and few headings or graphics. You might look at a legal court filing, a companys privacy statement, or an apartment lease. How could you redesign the...
-
On August 31, 2015, Harvey and Margaret, who file a joint return and live in Charleston, South Carolina, sell their personal residence, which they have owned and lived in for 10 years. The realized...
-
Which is an example of an indirect labor cost? a. Wages for gardeners and landscapers b. Salaries for F&B managers c. Wages for front desk agents d. The Executive Housekeepers monthly salary
-
Explain how you would find the value of a bond futures option.
-
Sally and Charles Heck received the following Form 1099-DIV in 2018: The Hecks also received the following dividends and interest in 2018 (Forms 1099-DIV not shown): Assuming the Hecks file a joint...
-
Adjustment for Uncollectible Accounts Below is the aging of receivables schedule for Evers Industries. Aging-of-Receivables Schedule November 30 Not 1-30 31-60 61-90 Over 90 Past Days Days Customer...
-
Figure shows a uniform beam subject to a linearly increasing distributed load. The equation for the resulting elastic curve is w. (-X* + 21X - L*X) 120 EIL Based on the above equation determine the...
-
Will either of the following reactions take place as written, according to the data inTable? (a) HCN + CH3CO2 Na+ Na* "CN CHCO2H Na* "CN CH3CH20- Na* |(b) CH3CH2OH HCN
-
What is the Ka of HCN is its pKa = 9.31?
-
Consider the following set of samples obtained from two normally distributed populations whose variances are equal: a. Suppose that the samples were independent. Perform a test of hypothesis to...
-
Consider the following income statement: Sales $ 875,904 Costs 569,856 Depreciation 129,600 Taxes 21 % Calculate the EBIT. Calculate the net income. Calculate the OCF. What is the depreciation tax...
-
Develop a Fortran program that simulates the motion of a pendulum using numerical methods. Create a Fortran subroutine that solves a quadratic equation given its coefficients.
-
If the returns on Stock A are as follows: Year 1 return 37 %, Year 2 return-_8 %, Year 3 return 12 %, Year 4 return-12 %, and Year 5 return-7 %, what is the average return for Stock A over this 5...
-
How do bacteriophages contribute to the dynamics of microbial ecosystems, and what potential applications do they offer for targeted antibacterial therapy and biotechnological advancements?
-
What are the key purposes of marketing research?
-
Why has the common-law doctrine of felony murder become controversial among courts and legal scholars in recent years?
-
Research corporate acquisitions using Web resources and then answer the following questions: Why do firms purchase other corporations? Do firms pay too much for the acquired corporation? Why do so...
-
a. Derive Eq. 7.4-8. b. Derive Eq. 7.4-12. c. Obtain an expression for the fugacity of a pure species that obeys the van der Waals equation of state in terms of Z, B = P b/RT, and A = aP/(RT) 2...
-
Draw structures for these compounds. (a) 2-Methylcylohexanol (b) 4-Methyl-4-penten-2-ol
-
Name thesecompounds: a) CHOCHCH3 OCH, b) CI
-
Name these compounds: (a) CH 3 CH 2 CH 2 NH 2 b) NHCH,CH,
-
Provide a project heading ( either at the beginning of page 1 or as a separate title page ) with the following: Project Management Plan Proposal Include two sections, as follows: Section 1 : Business...
-
Gatorade (a Pepsico product) is sold in each territory through competing independent wholesalers who then resell it to retailers. In contrast, All Sport (also a Pepsico product) is sold through a...
-
Your team writes up a list of reasons why Skype will help employees to not only become more efficient because they are being monitored, but also that using Skype to communicate with other employees...

Study smarter with the SolutionInn App


